There’s been a lot of fearmongering about chemical sunscreens. There were scary-sounding new stories – sunscreen absorbing into blood, killing coral reefs, zinc oxide being toxic, benzene in sunscreens…
Related Post: Top 7 Sunscreen and SPF Myths
I’ve covered most of these before, and they’re largely just the media taking things out of context and sensationalising them, partly for views, because a lot of media do survive off ad revenue and affiliate income now (…which is very different from me…).
Today’s topic – the EU has been reviewing the safety of sunscreen ingredients, and they’ve lowered some of the allowed limits, which means that some sunscreens currently on the market, both in the US and the EU, are over the “safe” limit.
Should you be scared? Should you change sunscreens? Does this prove US regulations are “unsafe”?
The video is here, keep scrolling for the text version…
EU limit changes
As mentioned, the EU has been lowering some sunscreen ingredient limits. For example, oxybenzone was allowed in sunscreens at a maximum of 6%.
But the SCCS have now said: “The use of benzophenone-3 [oxybenzone] as a UV-filter up to a maximum concentration of 6% in sunscreen products, either in the form of body cream, sunscreen propellant spray or pump spray, is not safe for the consumer.”
They now recommend a maximum of 2.2% in body products (6% is still the maximum recommended for face products).
So some people (including scientists from other fields) saw this and started making bold statements about how US sunscreens with 6% are unsafe, and telling people who are a bit more familiar with this area to “log off” because they’ve taken this one sentence very literally.

To be fair, if you only read this sentence, it does sound a bit scary. So let’s dive into why you probably don’t need to freak out, and what you might want to do based on this info.

A disclaimer: This is all going to be very simplified, I am not a toxicologist, but some toxicologists did help me out with the script.
Sunscreens in the EU
In most of the world, sunscreens are cosmetics not drugs, and this is the case in the EU. There are less strict rules about cosmetics than drugs, but the UV filtering ingredients that help sunscreens screen out the sun are still in a special category.
Their safety gets checked a lot more than other ingredients, and you can’t use them in sunscreens until they’ve been added to this special list (Annex VI). This also tells you the rules for using them, including the maximum percentages.
When it comes to sunscreens, and beauty products in general, people are always comparing the EU and the US, and one of the biggest differences is the sunscreen ingredients. But the situation is actually kind of the opposite of what you tend to hear – the EU allows a whole bunch more sunscreen ingredients than the US.
The US list hasn’t changed since 1999, when avobenzone was added. They only have about 7 commonly used “chemical” sunscreen ingredients:

The EU has about 20 common ones, including all 7 from the US. For these, the two regions had similar maximum percentages… until 2021 when the EU started shaking things up.
SCCS Safety Assessments
One thing I love about the EU is that they show their working, and it’s relatively easy to follow – it’s in these SCCS Opinions.

These “opinions” sound a bit more handwavy than they actually are. They’re *scientific* opinions, based on interpreting evidence according to almost 200 pages of guidelines.

But the word “opinion” is important – we’ll come back to that later.
They’re written by the Scientific Committee on Consumer Safety (SCCS). They focus on checking the safety of consumer products (these self-explanatory names are putting me out of a job…).
They look at sunscreen and other personal care products, but also household products like detergents, toys and clothes, and services like tanning and tattooing.
Their members are mostly toxicologists who specialise in risk assessment. There’s also chemists, pharmacists and dermatologists. Their very impressive CVs AND conflicts of interest are all publicly available, if you want to stalk them.
Every once in a while – if there’s important new data, or someone’s invented a new sunscreen ingredient – the European Commission gets the SCCS to look at potential risks and write an opinion.
These opinions include recommendations for using the ingredient, e.g. below a set percentage, or only in certain products (e.g. not on the lips). They might even recommend banning an ingredient altogether. Lawmakers will look at these recommendations, and most of the time, put them in the cosmetics regulation that EU products have to follow.

In late 2018 the European Commission decided to take a closer look at potential endocrine disruptors. This included some sunscreen ingredients, which is how this opinion came about – “The use of BP-3 up to 6% is not safe for the consumer,” but 2.2% is safe.

Not safe vs “not safe”
Let’s take a closer look at the word “safe”.
First off, the phrase “not safe” as used by toxicologists in a safety assessment doesn’t mean the same as what we would consider “not safe” in everyday life.
Scientists use words in weird ways – it’s just part of who we are, I’m sorry – and this can lead to a lot of confusion.
One example is the word “significant”.
In science it’s to do with how convincing the data is, so if I’m “significantly late” in a scientific study, I might be very consistently 10 seconds late.
But in everyday life, “significant” is about how important something is. So if my friend says I’m significantly late, I’m probably half an hour late, and they’re really pissed at me.
With the idea of “safe”:
In everyday life, we usually think of things as either “safe” or “harmful”.
If something is “safe”, it’s not going to hurt you.
If something is “not safe”, then there’s a good chance it will.
So this statement from the report kind of sounds like 2.2% oxybenzone sunscreen is fine, but if you use 2.3%, you’re going to pee blood, or grow a third arm, the worms in your eyelashes are going to make unsustainable farming choices…
But if there’s good evidence that terrible things happen at 2.3%, it would be supervillain-level irresponsible to let people to use 2.2%.
What if you use a little more sunscreen than you should? What if you scrape your skin and extra sunscreen gets in? What if some water in the sunscreen evaporates and that 2.2% gets a bit more concentrated? What if it turns out one of the pieces of data they used wasn’t quite right? What if a new study comes out?
Related Post: How to Reapply Sunscreen Over Makeup
So there is a big buffer put in when setting these “safe” percentages.
We can think of it more as a spectrum:
There’s a region where scientists can confidently agree something would not be harmful based on current info.
There’s also a region where scientists can confidently agree something would be harmful based on current info.
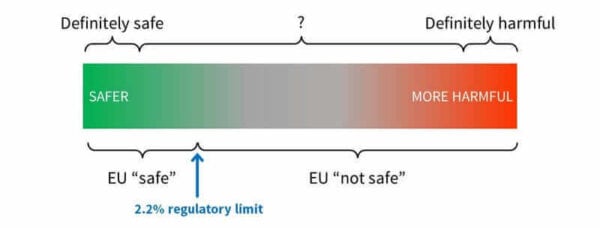
But there’s also this grey area in between, because in reality, there’s always some level of uncertainty and variation – people can use products differently, and respond to ingredients in different ways; studies don’t always translate easily to actual human use, new data can come out.
So “not safe” in the safety assessment doesn’t necessarily mean harmful. It means further in the grey zone than the EU has decided they’re comfortable with. This is one of the places where that “opinion” part comes in – different regions can have different approaches to working out their limits.
But how far is that current US/previous EU limit of 6% along this spectrum?
To find out, we can read… the rest of the SCCS report.
Hazard vs Risk
If you remember my post on clean beauty, how safe or dangerous something is – the risk – depends on both hazard and exposure.
Related post: Clean Beauty Is Wrong and Won’t Give Us Safer Products
A hazard is something that’s potentially harmful, like a lion, or even a rabbit – you could trip over it, it could convince you to do a whole bunch of messed up stuff.

But even a lion isn’t always going to be harmful. If it’s very far away in a cage, we have practically no exposure to it, so even though it is hazardous – it has that potential – the actual risk of harm to us is very low.
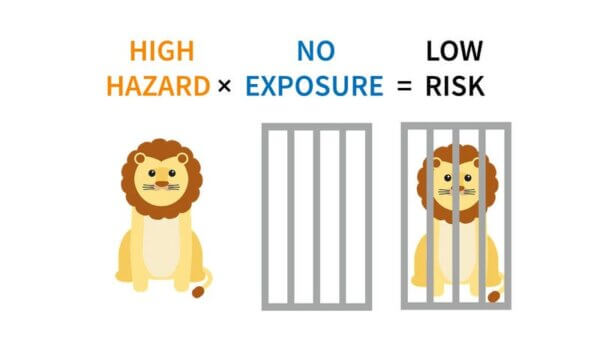
If our exposure is high – we’re in the cage with the lion – then the risk changes, and becomes very high, even though it’s the same lion.
This is that whole “the dose makes the poison” thing.
So for a safety assessment of a cosmetic ingredient, the SCCS will look at both exposure and hazard, to work out the overall risk – they put that lion in context.
Exposure
For exposure, the SCCS worked out how much oxybenzone 95% of consumers would be reasonably exposed to. So they try to capture normal use – if you’re eating sunscreen on toast every morning, it’s not going to be as relevant for you.
For most ingredients in beauty products, the main way we’re exposed is by absorbing it through skin. Sometimes there are other ways it gets into your body, like breathing in a spray, or eating some lip balm.
Here are some of the factors they take into account:
- Chemical properties
- Skin absorption data
- Product types
- Product usage (wash-off, leave-on, spray etc.)
- Usual concentrations in products
- Product amount
- Frequency of use
- Surface area
- Aggregate exposure
Most of these are probably things you expect – if you have an ingredient that absorbs easily through skin, you’ll have a higher exposure. It’s also higher if it’s in a leave-on product, if it’s used in products at high percentages, if it’s in a product that gets used in large amounts, or more frequently.
I want to highlight aggregate exposure – this is if the ingredient turns up in more than one product that you use. Every time I post about toxicologists doing safety assessments, someone will ask, “If it’s in more than one product, couldn’t it add up?” Yes it could, and toxicologists do take this into account.
For the oxybenzone calculation, they used 9.9% as the fraction of the total applied that would be absorbed into blood.
They estimated this from a study using skin samples, but they weren’t quite satisfied with the methodology, so they took the average absorption, which was 3.1%, and added two standard deviations to it to get 9.9%.
So the number they’re using is more than three times the actual average from the study – most people would probably absorb a lot less.

Related post: The “60% of products absorb into your bloodstream” myth (with video)
So you can see how they’re quite conservative with their estimates – whenever there’s uncertainty, they usually add extra buffer.
To work out how much oxybenzone was put on the skin in the first place, they used the average sunscreen application amounts from a couple of studies, which was 18 g of body sunscreen.
(Now some of you are probably typing in the comments – I apply much more than 18 grams on my body, that’s what the guidelines say, I could be using a dangerous amount – this will make sense in a bit!)
From this info they calculate an SED or systemic exposure dose, which means how much of the ingredient you would be exposed to in a whole body sense. They did this for an average 60 kg person, using those average amounts of a sunscreen with the maximum 6% oxybenzone.
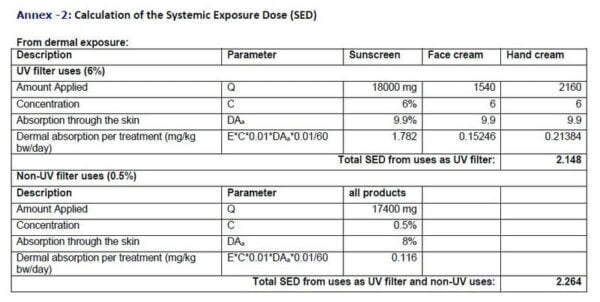
So for example, for body sunscreens, they calculated the SED to be 1.8 milligrams oxybenzone per kilo body weight per day.
Related Post: Purito Sunscreen and All About SPF Testing
Hazard
SCCS assessments also look at the hazard.

First they go through all the relevant data about the ingredient and how it could potentially be harmful. This includes things like skin irritation, allergies, cancer, fertility problems, damage to DNA, endocrine disruption.
They look at tests on cells (in vitro), tissues (ex vivo), animals (in vivo), and information from computer models (in silico). They also look at human data – for oxybenzone, there are some examples of clinical studies in volunteers, like the FDA studies from the “sunscreens absorb into blood” headlines.
Related post: More Sunscreens in Your Blood??! The New FDA Study
On top of this, they look for examples of people actually being harmed by that ingredient (case reports, post-marketing surveillance), and if there’s population-level data that’s been analysed to find links (epidemiological studies). They also look at information on similar ingredients that could be extrapolated (read-across).
Based on all of this, they pick a dose that they expect to cause little to no harm. For most cosmetic ingredients including oxybenzone, they use something called the No Observed Adverse Effect Level (NOAEL) – the highest amount of the ingredient that didn’t cause a harmful effect in studies.
Now, none of the chemical sunscreens commonly used in the EU or the US have been recorded to cause harm in humans, outside of things like allergic reactions and irritation, which can happen with pretty much any skincare ingredient.
But that’s not what we care about. We want to know about the longer term health effects that organisations like the EWG keep claiming our products are causing. Cancer! Endocrine disruption! Birth defects!

I’m kind of joking – but at the same time, these are the health effects that freak us all out the most.
Related post: Nail Polish, Miscarriages, Cancer? Not So Pretty Episode 2 (with video)
So for chemical sunscreens, there’s not a lot of human data. This is a good thing, because it’s real world evidence that there’s been no clear serious health effects in all the millions of people using oxybenzone since it was launched in the 1970s. But at the same time, for the purposes of a safety assessment, it’s less good, because it means you either have to:
A. feed oxybenzone to people until something bad happens, and scientists aren’t allowed to do this anymore except in movies, or
B. you have to translate information from less relevant studies (mostly animal studies).
Animals are similar to humans in many ways, but they aren’t the same. So scientists have to convert what happens in an animal to what happens in a human, and how accurate this translation is can vary a lot depending on the ingredient and the animal – chocolate and rat poison are both toxic to dogs, but I’m not going to stop eating chocolate, and I’m definitely not going to start eating rat poison.
So this approximation adds another layer of uncertainty.
(And this is another place where “scientific opinion” comes in. There can be different approaches to interpreting each piece of data, with different pros and cons, and a lot of the time, more than one approach is considered scientifically legitimate.)
For oxybenzone, the SCCS picked 14 endpoints from mouse and rat studies – 14 harmful effects that could be relevant to humans.
Then out of these, they picked the one with the lowest NOAEL (no-observed-adverse-effect level). This was from a study on pregnant rats being fed oxybenzone.
In this study, when pregnant rats were fed a lot of oxybenzone (1798.3 mg/kg body weight per day), they saw a whole bunch of stuff happen to the baby rats, including significantly reduced AGD, which is the distance between the, um, peach pit and the cherries – some scientist had to use a microscope to measure this, which I think is a pretty good setup for a joke. The full list:
- Significantly reduced anogenital distance (AGD)
- Reduced spermatocytes
- Delayed follicular development
- Reduced alanine aminotransferase (ALT)
- Significant increase in cholesterol levels
- Reduced body and organ weights
No change in:
- Litter numbers
- Number and weights of live fetuses
- Sex ratios
When rats were fed less (but still quite a lot: 207.1 mg/kg bw/day), they only saw one potentially bad effect – less sperm-producing cells in male rat fetuses.
So the SCCS picked one dose lower from the study for the NOAEL (67.9 mg/kg bw/d).
Margin of Safety
OK so now that we have these two numbers:
- The lowest “safe” exposure level from relevant animal studies (NOAEL), and
- the actual exposure consumers get (SED)
We’re ready to do the margin of safety calculation. Basically, how much are we getting with normal sunscreen use, compared to this “safe” amount?
The first row of this table shows that the margin of safety for a body sunscreen is 38. So in other words, if a 60 kg person applied 18 g of 6% oxybenzone sunscreen to their whole body, and absorbed about 1/10th of it, they would be expected to get 1/38th of the “safe” amount (NOAEL).
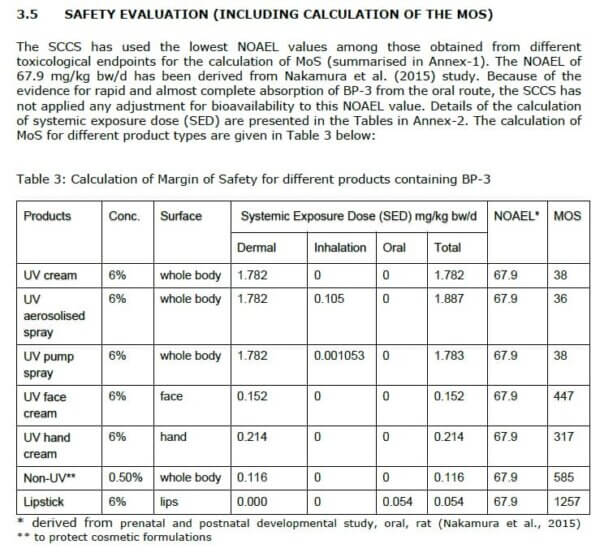
But the SCCS wants a margin of safety of 100, to take into account uncertainty – things like converting between rats and humans, if some people are more vulnerable than others, incomplete data. They want the average person to only get 1% of the “safe” amount (NOAEL).
So that’s why they dropped the percentage allowed from 6% to 2.2% – that’s how much would give the 60 kg person 1/100th of the “safe” (NOAEL) amount. (Note: they used the values for aerosolised spray where you might also inhale some sunscreen to calculate the new percentage – Row 2 of the table.)
If you wanted to actually get the “safe” amount (NOAEL), you’d have to use 100 times the average amount (1.8 kg a day – which I don’t think even sunscreen nerds do), or the average person would have to use sunscreen with 220% oxybenzone.
Now keep in mind that on top of this margin of safety of 100, there are a lot of other safety buffers here. For example:
- The effect they care about is a drop in sperm cells in a male rat fetus. Developing fetuses are a lot more sensitive than children or adults. The vast majority of people using sunscreen aren’t pregnant.
- The amount that actually caused that bad effect in the study (LOAEL = 207.1 mg/kg bw/day) was 3 times higher than the “safe” amount they used in the calculation (NOAEL = 67.9 mg/kg bw/day).
- And as I mentioned earlier, the amount actually absorbed in the skin absorption study was 3 times lower than what they used in the calculation.
So if you’re wondering how much you’d need to use to actually have the smallest harmful effect, based on the same data and assumptions, the sunscreen would have about 2000% oxybenzone – for a harmful effect on a male fetus, assuming it’s the same for humans and rats.
It’s also worth mentioning that the scientists who did the rat study didn’t think the sperm cell effect was that important – the “safe” level they indicated they would’ve picked was about 10 times higher.
Now I’m not saying that I disagree with the SCCS – they’re deciding what’s safe for a population, including people who are pregnant with a male fetus (although… probably not a male rat fetus?). And if you’re deciding for a population, with millions of people, then a tiny decrease in health risk can make some difference.
On top of that, there are tons of other sunscreen ingredients available in the EU – most European sunscreens don’t even use oxybenzone anymore, so it isn’t like sun safety will be massively impacted by this change.
But at the same time, you can see how cautious they’re being. There’s the massive margin of safety, the buffers they add whenever there’s uncertainty. On top of that, the new limit comes from new data from an animal study – there’s no evidence that 6% or 600% would actually be harmful in humans.
So “not safe” according to an EU safety assessment really doesn’t mean that you should panic if you’ve been using 6% oxybenzone sunscreen the whole time. Even though the EU is known for being really conservative when it comes to safety, it still isn’t like EU regulators have gone around knocking 6% oxybenzone sunscreens out of people’s hands, and they haven’t made any public announcements telling everyone to throw out their sunscreens.
The report was published in March 2021, the limits came into place in July 2022, then companies have another 12 months to stop selling products that are over the limit. That’s 2 and a bit years between the finding of “not safe for the consumer” and no longer having the products available. So this really highlights that “not safe” and “harmful to your health” are very different.
So that’s for body products – why is the limit different for face products?

As you might’ve guessed from the explanation, you apply a lot less of a face product, on a smaller area of skin. The SCCS found a margin of safety of 447 for a face cream with 6% oxybenzone, so that was confirmed to be “safe”. This was for an application amount of about a quarter teaspoon (1.54 g), so you could apply that 4 times a day and still stay under that 1% margin of safety.
Other sunscreen ingredients
Let’s take a quick look at the SCCS safety assessments for the other sunscreen ingredients.
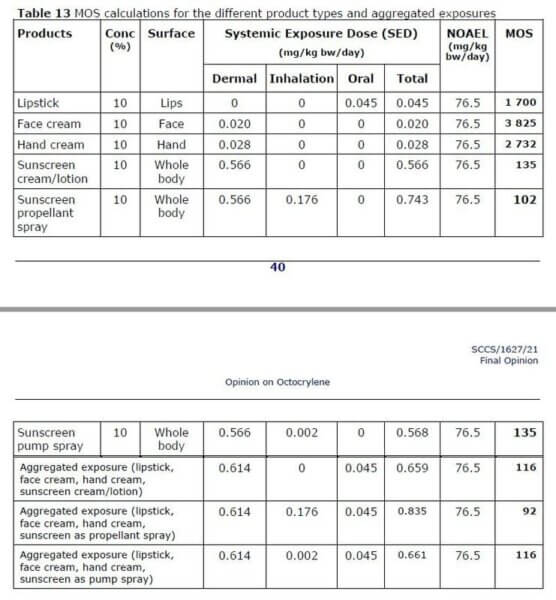
Octocrylene was confirmed as “safe” at up to 10% for humans. The exception was spray products when used in combination with other products (taking into account aggregate exposure). Then it would have a margin of safety of 92, so the limit was lowered to 9% to get it back to a margin of 100.
Homosalate was found to be “not safe” at 10%. The limit was lowered to 0.5% for body products, and 7.34% for face products. This is actually a bit of a problem for body sunscreens, because homosalate is useful for keeping other sunscreen ingredients dissolved, so there’s going to be a lot of reformulating.
For both of these, the SCCS found there wasn’t enough evidence to say they’re endocrine disruptors.
For 4-methylbenzylidene-camphor (4-MBC), the 4th sunscreen ingredient assessed, the SCCS said there was enough evidence that it “may act as an endocrine disruptor and has effects on both the thyroid and estrogen systems”. There wasn’t enough data to work out what the new limit should be, but they did say it would be lower than the current limit of 4%.
Octinoxate and titanium dioxide will also be reassessed soon.
What should you do?
If you’re a cautious person, you might be wondering if you should switch to sunscreens that follow these new limits, or even avoid these ingredients completely just in case.
I can’t give you personal advice – everyone’s risk calculus is different, it’s really a conversation you should be having with your doctor.
Related Post: Top Sunscreen Recommendations 2022
But generally speaking, there are some useful takeaways from these safety assessments that are worth considering when working out your personal pros and cons:
Dose
Dose – how much sunscreen you use and how often – is important. The SCCS assessment arrived at their limit based on using 18 grams a day, but you might be applying a lot more or a lot less.
For example, I only apply full body sunscreen a few days a year when I go to the beach, but someone who works outdoors might be applying and reapplying several times a day, most days.
Related Post: How Do Sunscreens Work? The Science
Vulnerability
Vulnerability is also important. I’m not talking about sharing your childhood trauma to do well on a reality TV show – it’s whether the risks of a particular sunscreen ingredient are especially relevant to you.
The most common concern is probably pregnancy. A developing fetus is more strongly affected by all sorts of things (alcohol, aspirin, camembert cheese…). As we saw, these safety assessments do look at what happens to developing fetuses, and a lot of limits are set based on that. But if you’re pregnant, you would have more reason to consider temporarily lowering your exposure to risks, even just for peace of mind.
The other side of the equation is also important – what are the costs of not using that sunscreen ingredient?
You might consider things like:
- a family history of skin cancer
- how much UV exposure you get
- whether or not you have access to suitable alternatives. Are other sunscreens available where you are and within your budget? Will stretching your budget make you underapply? Is protective clothing appropriate for whatever you’re doing?
I’ve talked about hats, umbrellas and UV protective clothing before.
Related posts: Do Hats and Umbrellas Protect Well From the Sun? (with video) and How to Choose UV Protective Clothing
I think it’s also worth noting that safe limits for cosmetic ingredients will be constantly changing, based on new data, or new regulations, or new procedures for working out safety.
There is always going to be some uncertainty. And I know at a gut level it does feel uncomfortable – humans haven’t really evolved to be OK with uncertainty when it comes to potentially harmful things.
But it’s worth remembering that everything is potentially harmful. We just don’t necessarily think of them that way because we don’t see announcements about new risk assessments for most of them.
And these adjustments are actually a good sign that the system is working the way it was designed, rather than a sign that something has gone wrong. The vast majority of the time, these changes have happened because of new data from models and experiments, not because people have gotten hurt from using products with these ingredients.
It’s a reminder that there is research happening, there are experts putting new research into context against all of the other evidence, and updating guidelines regularly, to make products that are already pretty darn safe, even safer.
Thanks to Mo Skin Lab, F Anthony Simion PhD, ERT (University of Cincinnati) and Jay Gooch PhD for assisting with this post! All mistakes remain my own.
References
Scientific Committee on Consumer Safety, SCCS Notes of Guidance for the Testing of Cosmetic Ingredients and their Safety Evaluation 11th revision, 2021
Scientific Committee on Consumer Safety, Opinion on Benzophenone-3, 2021
Scientific Committee on Consumer Safety, Opinion on Octocrylene, 2021
Scientific Committee on Consumer Safety, Opinion on Homosalate, 2021
Scientific Committee on Consumer Safety, Scientific advice on the safety of Homosalate as a UV-filter in cosmetic products, 2021
Scientific Committee on Consumer Safety, Opinion on 4-Methylbenzylidene camphor (4-MBC), 2022
Nakamura N, Inselman AL, White GA, et al. Effects of maternal and lactational exposure to 2-hydroxy-4-methoxybenzone on development and reproductive organs in male and female rat offspring. Birth Defects Res B Dev Reprod Toxicol. 2015;104(1):35-51. DOI:10.1002/bdrb.21137











Goodness, I am glad you are wading through all this information, I could never do that and understand it all and I have a degree in biology and understand a lot of the stuff about dose response and so on. It does show how difficult (impossible?) it is for the average person to work out for themselves what it all means. We do need to trust the regulators and try and disregard the fear mongers, click bait and vested interests around us. (much easier said than done) Thanks Michelle.
I agree, it is a good thing that the regulations change as it means that the things keep being looked at, even though it feels scary when they do and you start worrying about what you have done until then.
Thank you for sharing the facts, it must have been a lot of work to go through all that data.
You haven’t taken into account that octocrylene containing products produce benzophenone over time. ConsumerLabs has done an in-depth analysis of all the research and warns against the use of octocrylene – all chemical sunscreens actually.
Talked about benzophenone and octocrylene here: https://labmuffin.com/octocrylene-causes-cancer-and-im-propaganda-now-with-video/ Unfortunately it looks like Consumer Labs just repeated what the study said without much critical analysis.