What is hyaluronic acid and how does it work in skincare and makeup?
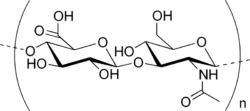
What Is Hyaluronic Acid? Hyaluronic acid (HA), along with its cousin sodium hyaluronate, is an immensely popular ingredient in skincare and makeup. It’s also naturally found in the body. Most of it dwells in the extracellular matrix, the scaffold which holds up the cells of your skin. Hyaluronic acid is found in both the epidermis and the deeper dermis, where …