Over the past two years, my post on the chemistry of micellar water with dodgy photographed scrawlings has become one of the most popular, so I thought it was high time to update it with nicer drawings and finetune the explanation of the science.
This also comes in video form – check it out here!
There are tons of micellar waters on the market now, many of them with similar active ingredients.
The essential ingredients in any micellar water are:
- Water (obviously) and
- One or more surfactants.
Surfactants are cool ingredients that I’ve written about a lot (including in my guest post on The Toast). They’re useful molecules with a hydrophilic head that’s attracted to water (and repels oil), and a lipophilic tail that’s attracted to oils and grease (and repels water).
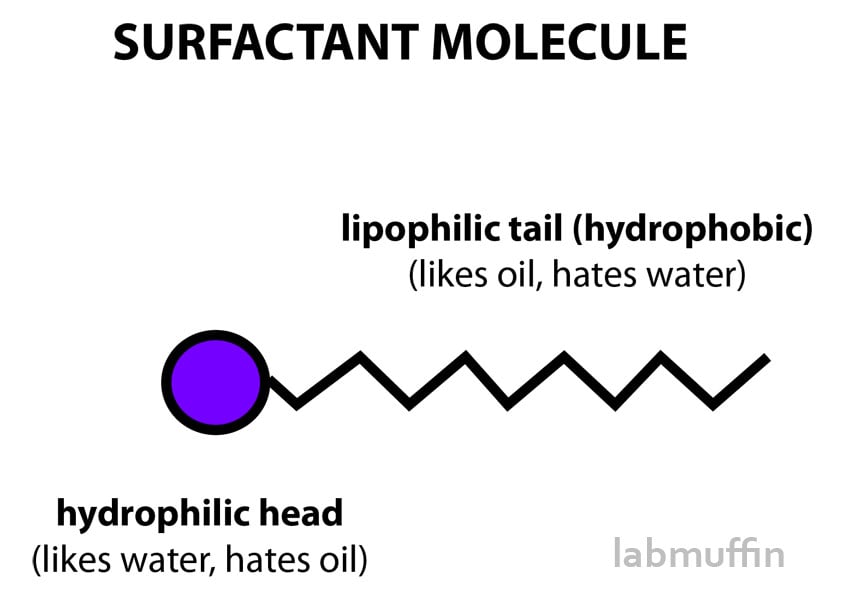
Oil and water normally repel each other, so they try to stay away from each other. This means that oil doesn’t dissolve in water (which you’ll know if you’ve ever tried to wash an oily dish), and instead sits on top like a bad toupee.
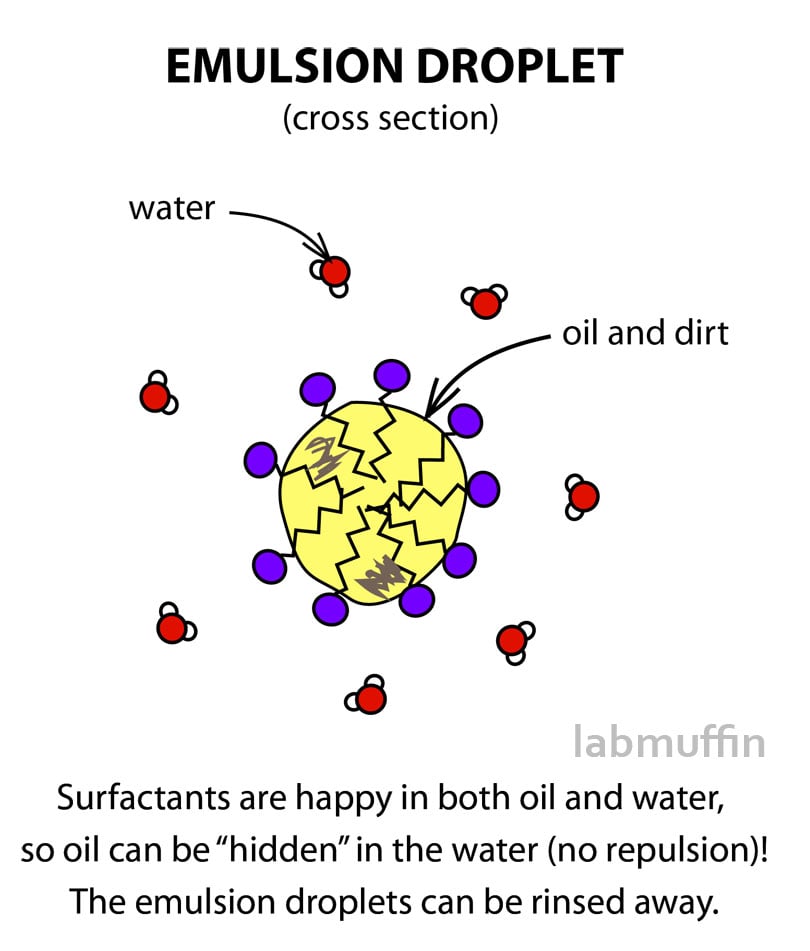
When surfactants are added to an oily dish, for example, and then scrubbed with water, they help it break up into droplets (emulsion droplets). They surround the oil and “hide” it from the water, allowing it to be smuggled out and washed away to leave a clean surface.
Surfactants are the key ingredients in micellar water, as well as in detergent, soap, shower gel, face wash, shampoo and so on. You’ll also find them keeping oil and water happy together in emulsion products like moisturisers and mayonnaise.
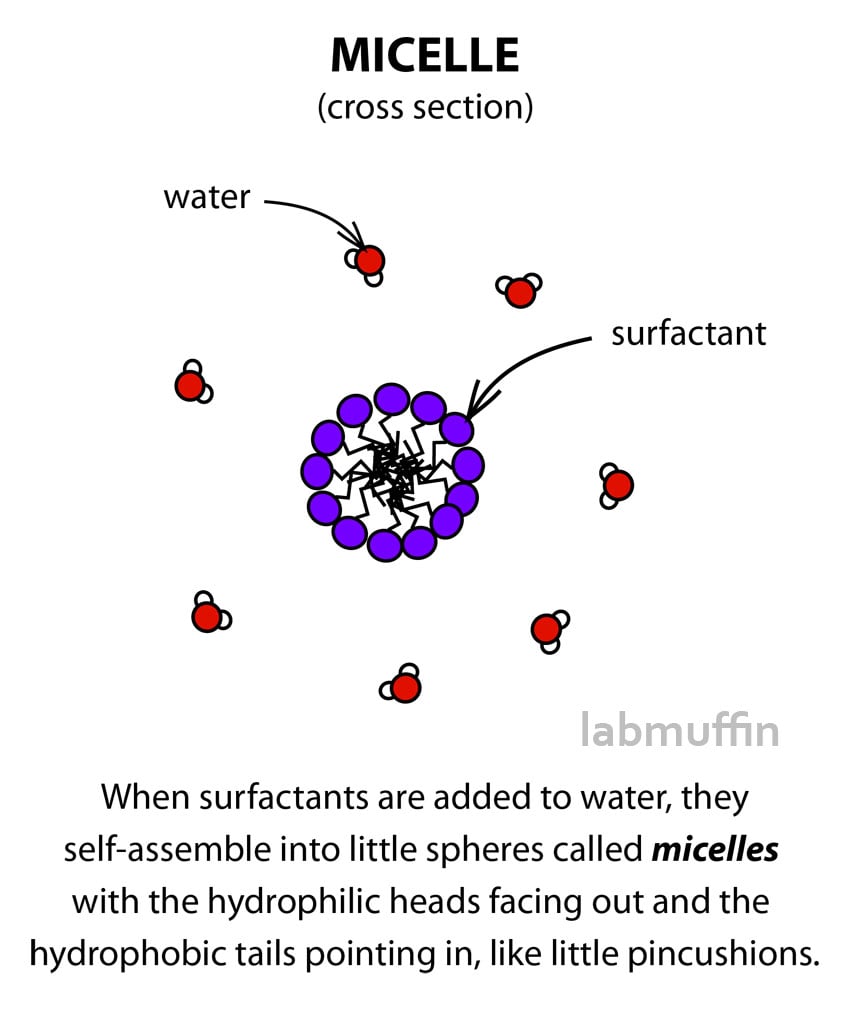
So let’s get back to micellar water. When enough surfactant is added to water (more than something called the critical micelle concentration or CMC), the surfactant molecules assemble themselves into clusters called micelles.
These micelles are spherical arrangements of surfactant molecules, with the tails pointing in and the heads facing out – this means the hydrophobic tails are protected from the water by the hydrophilic heads.
Some brands of micellar water contain oily substances, like Nivea Sensitive 3-in-1 Cleansing Water which contains grape seed oil. In these products, the oily substance will sit in the middle of the micelle, like in the emulsion droplet.
(In my previous post, I labelled the emulsion droplet a micelle – I’ve been told by a materials chemist I work with that that’s incorrect.)
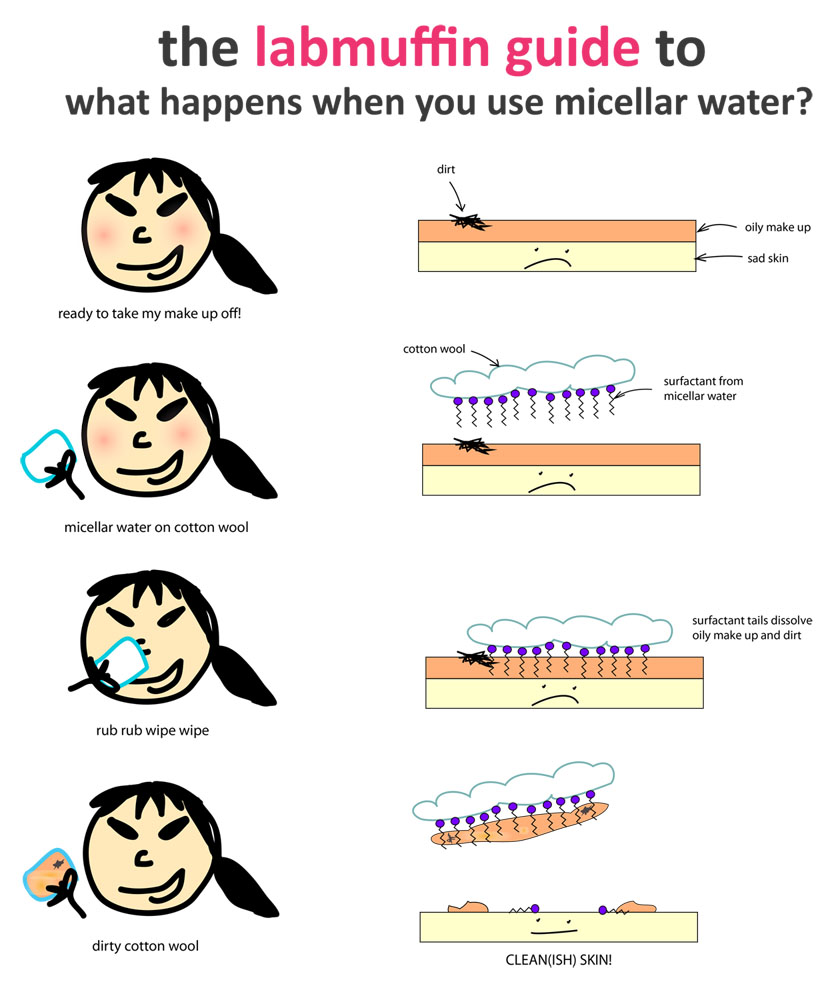
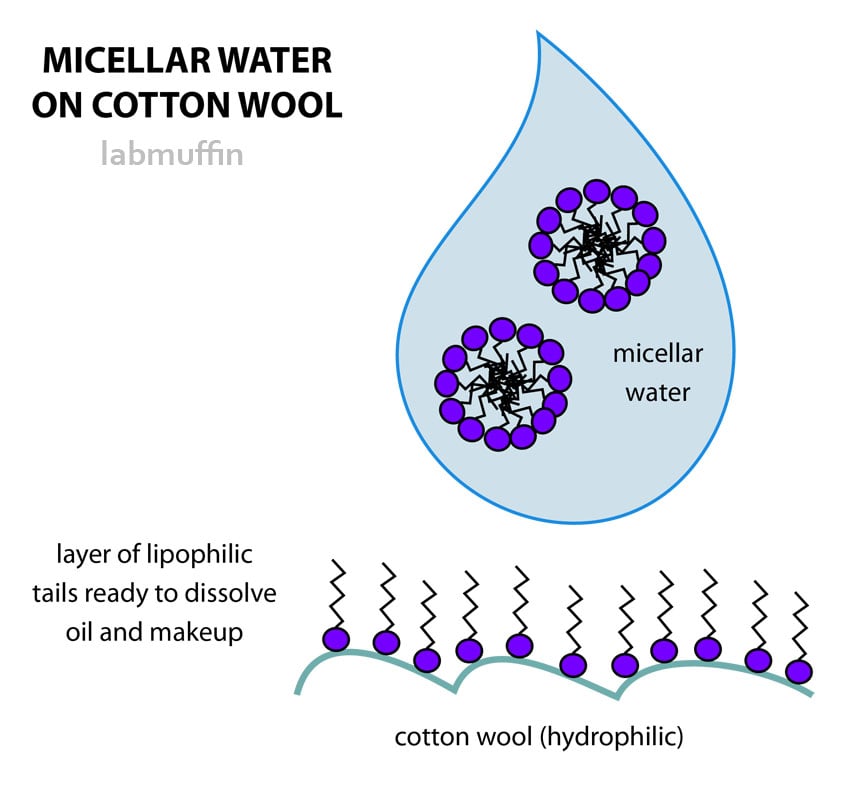
 The micelles aren’t bound together into a molecule, which means they’ll rearrange easily. If the micellar water is poured onto a cotton wool pad, for example, it rearranges so that the heads are stuck to the cotton wool (made of cellulose, which is hydrophilic), and the tails stick out (the technical term for this is an oriented monolayer).
The micelles aren’t bound together into a molecule, which means they’ll rearrange easily. If the micellar water is poured onto a cotton wool pad, for example, it rearranges so that the heads are stuck to the cotton wool (made of cellulose, which is hydrophilic), and the tails stick out (the technical term for this is an oriented monolayer).
This means there’s a neat oil-loving layer sitting on your cotton wool pad. Since make-up, sebum, grease and dirt are all oily, this means it’s perfect for cleaning your face at the end of the day.
Here’s what it looks like on a microscopic scale (diagrams are really really not to scale):
 When you wipe off the make-up with the cotton pad, the layer of lipophilic tails absorb the oily make-up. Since it’s only one layer of tails, you might need to go through a few pads to get rid of everything if you’re wearing thick make-up or you’re super greasy. If you don’t rinse your face afterwards, you’re also likely to have bits of micellar water left on your face afterwards.
When you wipe off the make-up with the cotton pad, the layer of lipophilic tails absorb the oily make-up. Since it’s only one layer of tails, you might need to go through a few pads to get rid of everything if you’re wearing thick make-up or you’re super greasy. If you don’t rinse your face afterwards, you’re also likely to have bits of micellar water left on your face afterwards.
This leads to one of the most common questions I’m asked: Do you need to rinse your face after you use micellar water?
Sadly, the answer to this question is a non-committal, unsatisfying “it depends”.
Micellar water contains mild surfactants, and surfactants can disrupt the structure of the skin – sodium lauryl sulfate, for example, is a strong irritant. But not all surfactants are made equal.
Like how some people can’t tolerate any sodium lauryl sulfate on their skin while others can use it daily for years with zero problems, some people will be able to tolerate the surfactants in micellar water better than others. And just to make it even more complex, there’s a range of surfactants used in different micellar water products, with different irritancy potentials.
If your skin feels dry or itchy after using it, I would definitely recommend following the micellar water with a wipe of a cotton pad soaked in plain water.








The only thing I love more than your blog, is this post. Yay surfactants! I’ve always found with micellar water that I have to do at least 4 swipes per area to really get all the gunk off my face.
I wasn’t even aware there were micellars with SLS, which sounds CRAZY, because WHY WOULD YOU WANT TO LEAVE THAT ON YOUR FACE!?
xoxo
A
http://behindthescenes-beauty.com
There aren’t any micellar waters with SLS in them, but there are lots of facial cleansers with them, and one cream that used to be recommended for babies with eczema??? It was called Aqueous Cream. Crazy!
Most likely the real reason why it’s called Aqueous Cream, because the formula is taken from National Formulary or British Pharmacopeia and this is the name that is used in both of them. It’s probably TGA approved goods and Sodium Lauryl Sulphate is used as emulsifier to comply with regulations.
This post is all kinds of awesome! The pictures are so cute and effective in helping us understand the concepts. 😀
I’ve been using micellar water for a few years now but lately I find that having to wipe my face several times with a cotton pad has become kind of irritating on my skin. 🙁 I’m thinking it’s because I use Epiduo. I still love micellar water though hahaha 🙂
Thank you for this highly informative post!
This is an awesome post! I find I definitely have to use a cleanser after using Micellar water – I don’t like the way it makes my face feel leaving it on. Same with using face wipes, at the bare minimum I have to rinse my face.
I love this post! So explanatory of something that we really don’t think much about! Thank you for sharing 🙂
Love this post! Thanks for the SCIENCE! The diagrams are adorable.
I don’t know how some people can just use micellar water/make up wipe and end their cleansing routine. All that residue! I like micellar water because it doesn’t leave an oily film behind but I’m most definitely at least rinsing my face again, even if I don’t follow up with a cleanser.
Me too! Using wipes as my only cleansing step also feels totally weird for me. I only really use micellar water when I’m camping, or otherwise don’t have access to a tap, but I’m also a bit pedantic with my skincare routine so I expect there are lots of people out there for whom this isn’t a big deal…
Great Blog! Atlast! the blog that explains things the way they really are! Thank you for your time Michelle!
Not all micellar water contains detergents, some of them contain solubilizers,.
The ones with detergent-surfactants are need to be rinsed off, but the micellar water with solubilizers can be left on the skin as they have more skin-friendly ingredients and their principle is to dissolve the dirt on the skin surface without disturbing skin’s protective mantle.
Do you have any examples of solubilisers used in micellar waters that aren’t detergents? The definition of a detergent is any non-soap synthetic surfactant, and I’m having trouble imagining a solubiliser that isn’t a surfactant unless it’s a straight-up solvent.
No, i don’t have examples that are on the market , sorry i don’t buy cosmetic products I create them. But, out of curiosity, i’ll google something for you when i have time, ok?
Anyway, “detergent” is not really right word /definition that i’ve used here…
Surfactant have very broad meaning for instance;
Surfactants are useful for the following application.
1. Cleansing
2. Emulsification
3. Solubilization
4. Conditioning
5. Special effects
Cleansing Surfactants
The useful thing about micelles is that they can help suspend oil in water. When a small amount of oily materials is put into an aqueous solution of surfactants, it will migrate into the center portion of the micelle. So, when you put a surfactant solution on a surface like hair or skin, the oil that is there will be drawn away from the surface and into the micelles. When the surfactant solution is rinsed away, the surface is clean.
Foaming
Foam is another characteristic of surfactant solutions so you’ll need surfactants if you want your product to foam. Essentially, foam is the entrapment of air in liquids and the alignment of the surfactant molecules helps keep the foam stable. It should be noted that foam itself is not related to the ability of a product to clean. But consumers expect cleansing products to foam so as a cosmetic formulator, you’ll have to add foaming surfactants.
Emulsification
While cleansing cosmetics remove oils, many cosmetic products are design to add oily materials to the skin and hair. These ingredients usually can’t be applied directly because they have undesirable aesthetic characteristics in their concentrated form. For this reason, cosmetic chemists create emulsions using surfactants.
In the simplest case, an emulsion formula is made by mixing an oil phase with a water phase and a surfactant. The micelles created by the surfactant entrap the oil in their centers and it remains suspended throughout the mixture. Products like creams and lotions are typically emulsions. When the product is applied to the skin or hair, the surfactant micelles break open and deliver the oily materials.
Solubilization
The problem with most emulsions is that they usually create opaque products. However, there are times when a cosmetic chemist wants a clear formula but still wants to blend an oil in a mostly water formula. Fortunately, there are surfactants that have the ability to create particles so small that light passes through them and the solutions remain clear. Molecules that do this are solubilizing surfactants. They are used to blend oily materials like fragrances or natural ingredients into clear solutions. An example would be a surfactant like Polysorbate 20.
Conditioning
Since surfactants often contain an “oily” part on their molecule, they have conditioning properties that can improve the feel and look of the surfaces of skin and hair. For them to work this way, the surfactants have to be left behind and also be non-irritating. This can be achieved through a leave-on cosmetic product or by using surfactants that can bond to surfaces through an electrostatic charge
Special effects
In addition to the four properties discussed, surfactants have a number of other special effects that are useful to formulators.
For example, some surfactants have anti-microbial effects so they can be used as a preservative. Certain surfactants can be used to create an elegant, pearly effect in cosmetics to increase their aesthetic appeal. They can be used for thickening systems, reducing irritation and improving formula stability.
Even an ingredient would be helpful! I think you might have confused detergents with anionic detergents? Anionics are generally more irritating, but others are too. Polysorbates for example are non-ionic detergents, and they will still irritate skin to some extent.
Thanks for this comment, it was really informative. I came from Reddit reading about MLMs and was pleasantly surprised to learn some chemistry.
Just to make things make sense in my mind, the detergent-surfactants you’ve talked about are similar to fatty acids in soaps, right? So you need to add water to do any cleaning. But these magical solubilizers are basically a watery cleanser that you can apply directly to the skin, bc they encase makeup oils in micelles that are removed with pressure (like wiping?) I don’t use makeup, so I’m not familiar with these things.
Are either vegetable glycerin or witch hazel considered surfactants? Saw a DIY recipe for micellar water, and was confused as to what was adding the micellar component to the water (if anything) in that recipe. Rose water, witch hazel, and vegetable glycerin, and the optional addition of some carrier oil…did not make any sense to me, but maybe I just don’t realize one of those is acting as a surfactant.
Nope, neither of them are surfactants! Glycerin can be a good solvent though.
Great post! But can micellar water effectively remove waterproof sunscreen?
I am also wondering this same thing!
Hi! I enjoyed this post on micelles and which one is worth trying! Skyn Iceland perhaps! I’m a nurse and I’m in school again to become an NP! Can I get an invite to join your blog? Thanks!
Thank you! I’m not sure what you mean by invite?
Aren’t the concentrations of surfactants in other surfactant-based cleansers (i.e. most cleansers) already well above CMC? What makes micellar water different? It sounds like just a gimmicky way of saying “water with surfactants.”
The surfactants used in micellar water tend to be less irritating than those in most other cleansers, so they can be used on the eyes and left on the skin for most people.
I see. Thanks for the response. I don’t expect you to have an answer to this, but I’d like to take this opportunity to wonder aloud why it’s marketed as “micellar water,” instead of something more descriptive, like “no-rinse cleanser.”
As a catchier name, I assume – the same reason we have serums and lotions and creams instead of heavy moisturiser, light moisturiser, moisturiser with more actives etc.
Thank you for this! I have been trying to find out if micellar water can be used effectively with something other than disposable cotton pads, but based on this, my impression is not – is this correct? Is there a reusable option that I don’t have to throw away, like a cotton or microfiber cloth? Thanks again!
Omg what is it? Wonderful post!
Oh God i really love your post, darling! It’s really easy to understand even for me. Did you do some science experience by yourself for this post or you did research with so many journal maybe? Oh, did you deal it with your dermatologist? it’s so great!
Omg what is it? Wonderful post!
Oh God i really love your post, darling! It’s really easy to understand even for me. Did you do some science experience by yourself for this post or you did research with so many journal maybe? Oh, did you deal it with your dermatologist? it’s so great!
Interesting fact-check on micellar water! I had no idea it was actually a type of cleanser. Thanks for the update, Lab Muffin! I’ll definitely give it a try. 💦🧖♀️