Much like “organic” and “all-natural”, “paraben-free” is one of those phrases you’ll see displayed prominently on increasing numbers of skincare and beauty products. What are parabens, what health effects do they have, and should you be avoiding them? Here’s the science behind the marketing.
(I’ve written a much simpler rundown of parabens here, if you want a quicker overview.)
What are parabens?
Parabens are a family of preservatives commonly used to control the growth of microbes in cosmetics, toiletries, food and pharmaceuticals. They are esters of para-hydroxybenzoic acid, a naturally occurring chemical found in many fruits and plants. Their chemical structures and actions are very similar, with the “R” group changing as shown below.
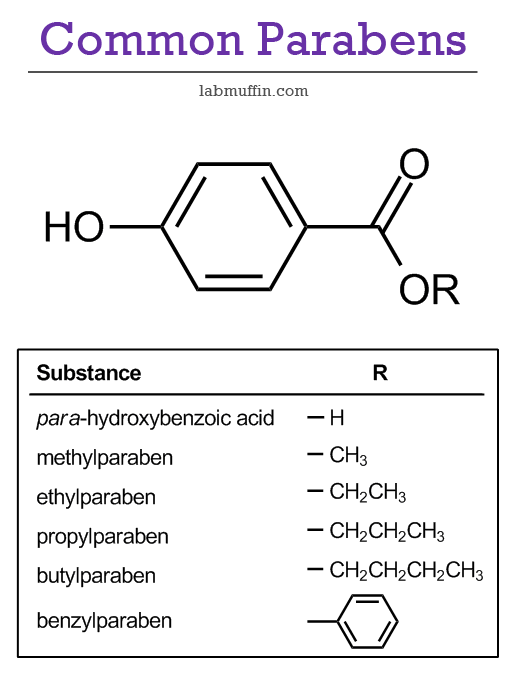
The most commonly used parabens are methylparaben, ethylparaben, propylparaben and butylparaben, although many others (isopropyl-, isobutyl-, pentyl-, phenyl-, benzyl-) have been used in products as well. Different parabens work best under different conditions and act against different microbes, so you’ll often see them used in combination to enhance the preservative effect.
Parabens were developed in the 1920s and these days, they’re the most widely used preservatives in cosmetics, appearing in over 85% of products. Parabens are popular for good reason: they’re inexpensive, effective in very small amounts, work well in most products, and act against a wide range of nasty microbes. They have a very long record (almost 100 years) of safe use. The only reliably linked harmful health effect is allergy, which occurs in a tiny fraction of people, and it’s often only a problem on broken skin.
Why do parabens have a bad reputation?
Despite all these advantages, parabens have become well known as a “nasty” in the last 10 years. This came about when a few studies appeared which led people to question whether parabens were really as safe as they seemed:
In 1998, Routledge et al. found that parabens were weakly estrogenic in rats – that is, they bind to estrogen receptors and can hence act as female sex hormones. Excessive exposure to estrogens has been linked to the development of breast cancer and reproductive disorders.
In 2004, the Darbre group reported that they found parabens in 20 different human breast tumours. This study boosted concern about parabens to an all-time high, with advocate groups like the Campaign for Safe Cosmetics, the Environmental Working Group and the David Suzuki Foundation (compilers of the “Dirty Dozen”) calling for bans on parabens in personal care products. In response, Denmark banned the used of parabens in products for infants in 2011.

The EU Commission (who haven’t banned parabens, contrary to all internet rumours) has limited the sum of propylparaben and butylparaben in products to 0.19% and banned their use in leave-on products designed for the diaper area of children under 3. They’ve banned the use of isopropyl-, isobutyl-, pentyl-, phenyl- and benzylparabens altogether not due to evidence of harm, but due to lack of data. The limits of 0.4% each for methylparaben and ethylparaben, and 0.8% total for all parabens have been unchanged since 2000.
A 2014 study, also by the Darbre group, reported that multiple parabens used together could cause the rapid growth of breast cancer cells in vitro (cultured outside of the human body).
Why haven’t parabens been banned?
At first glance, the studies seem to be concerning. However, most scientists and virtually all regulatory bodies who have reviewed these studies aren’t too worried about parabens. Here’s why:
The 1998 Routledge study showed that parabens are really, really weak
The Routledge study performed in vitro tests on cultured yeast cells, and in vivo tests on live rats. The parabens tested (methyl-, ethyl-, propyl- and butyl-) were found to be thousands to millions of times weaker than estradiol, the naturally occurring female sex hormone. Butylparaben, the most potent paraben used in the study, was 10 000 times weaker than estradiol in vitro and 100 000 times weaker in rats, while the least estrogenic paraben, methylparaben, was 2 500 000 times weaker in vitro and completely inactive in rats. It’s also worth noting that the study injected the parabens under the rats’ skin, which hopefully isn’t what you usually do with your cosmetics! None of the parabens were active when fed to the rats.

The 2004 Darbre study was flawed in a lot of ways
I was quite surprised when I read the Darbre study (aka “The study that started all the fuss”). There are a whole bunch of things that made me wonder how it managed to get so much traction. I’m blaming the media and advocate groups for being bad at interpreting scientific studies. It seems like it’s become a game of whispers, where the actual information gets increasingly distorted as it’s passed along. Here are some of the more glaring issues, which have been repeatedly pointed out by the numerous scientists who have reviewed the paper:
- No comparison with control (non-cancerous) tissue: The study reported finding parabens in breast tumours, without any comparison with normal, non-cancerous tissue. This is pretty meaningless because if you find parabens in both, it suggests that the presence of parabens isn’t linked to breast cancer. It’s like observing that everyone with cancer drank water before they got cancer – it doesn’t mean water causes cancer because everyone drinks water. And even if there were higher levels found in the tumours than the normal tissue, it still doesn’t necessarily mean there’s a causal link. Even for natural estradiol, which is much more estrogenic, there isn’t a clear link between its presence in breast tissue and cancer.
- Parabens were measured in blank samples: In the study, parabens were found in blank samples containing no tissue, which suggests that the equipment was contaminated with parabens. This was kind of understandable, since the amounts of parabens found were so small and parabens are everywhere… but it’s also a good reminder that the amounts found were very small. The quantities of parabens found in the breast tissue were nanograms per gram, which is on the scale of one in every thousand million, or half a teaspoon in an Olympic-sized swimming pool. This would be worrying if they were potent, but from the Routledge study, we know they aren’t. More bizarrely, some of the blanks contained more parabens than the actual samples. This raises the possibility that none of the parabens measured actually came from the breast tumours.
The commentary accompanying the paper talked a lot about how the study supported a potential link between parabens and breast cancer, but after a lot of criticism from other scientists, the authors published a bit of a backtrack:
“Nowhere in the manuscript was any claim made that the presence of parabens had caused the breast cancer, indeed the measurement of a compound in a tissue cannot provide evidence of causality.”
Unfortunately this was published 6 months after the initial study, and the media didn’t seize on this statement with anywhere near the same amount of enthusiasm.
There’s still no direct evidence that parabens have ever caused bad hormonal effects in humans
Parabens have been around for almost 100 years, are used everyday by billions of people, and there are lots of studies showing their safety. Yet there still hasn’t been a study showing a convincing link between paraben use and negative health effects, apart from reactions in those few unlucky people with paraben allergies. (There are a couple of epidemiological studies that looked at the use of antiperspirant deodorants and breast cancer, but these don’t usually contain parabens and the results weren’t clear cut. A couple of studies found an association between urinary paraben levels and some markers of oxidative stress, but this is far from a convincing link to cancer.) The studies that are frequently cited to support a link between parabens and endocrine disruption are in vitro and animal studies, both of which are difficult to link to effects in humans.
It’s impossible to take an in vitro result, where isolated cells are treated directly with the chemical of interest, and translate it reliably to a real effect in a living organism. In vitro studies are typically way more sensitive than in vivo studies on living organisms. Since chemicals in in vitro studies are placed directly on the cells, they can’t replicate how the chemicals will actually act inside a complex organism containing biological systems that can block, inactivate/activate and/or remove the chemical before it reaches the relevant cells inside the body. This xkcd comic is relevant:
The in vivo studies that showed that parabens had estrogenic effects were animal studies, primarily in rats. Obviously humans aren’t rats so a lot of substances don’t act the same way, much like how chocolate and xylitol are both fine (and beneficial) for humans, but not dogs. Additionally, very high doses were used in the animal studies, which don’t translate to the very small concentrations (typically less than 0.3%) used in cosmetics and personal care products. Things that are bad at high doses aren’t always bad at low doses; for example, drinking too much water leads to lethal hyponatremia. Additionally, the vast majority of the studies looked at feeding or injecting the parabens; very few of the studies actually involved applying parabens to skin.
There is one study on applying a cream containing 2% butylparaben to the entire bodies of volunteers for a week. Even though the cream contained way more paraben than a regular product (which generally has less than 0.3%), the researchers founds that butylparaben “did not seem to have any short-term influence on the levels of reproductive and thyroid hormones.”
We’re exposed to far more potent estrogenic chemicals that aren’t dangerous
Personal care products use very small amounts of parabens, and as discussed above, we know that they’re not very potent. The small amounts of parabens that get through the skin are mostly metabolised by skin enzymes into para-hydroxybenzoic acid, an antioxidant found in many plants and foods that’s even less estrogenic than parabens. This is then excreted through the urine. There isn’t any evidence that parabens accumulate in the body. A small amount of intact parabens seem to be able to make it into breast tissue though, and the 2015 Darbre study raises the concern that these parabens, in combination, could exert effects on breast cancer cells.
This seems quite unlikely if we consider what we know about other, far more potent estrogenic chemicals we regularly encounter, like ethinyl estradiol and phytoestrogens. Ethinyl estradiol, the estrogen found in combined oral contraceptive pills, is about 2 000 000 times more potent than butylparaben, and even then it’s only associated with a small increase in cancer risk.
Phytoestrogens are naturally-occurring estrogenic compounds found in foods. Soy phytoestrogens in particular have attracted a lot of attention as it contains the particularly potent daidzen and genistein, which are about 200 times as potent as propylparaben. This review calculates that exposure to 3000 times the normal amount of butylparaben would present less estrogen exposure than normal dietary phytoestrogen. However, not only are dietary phytoestrogens not associated with breast cancer, the current evidence seems to point towards them being protective against breast cancer. This review on phytoestrogens also makes an interesting point:
“although [phytoestrogens] behave similarly to numerous synthetic compounds in laboratory models of endocrine disruption, society embraces these compounds at the same time it rejects, often with vigor, use of synthetic endocrine disruptors in household products.”

Why not avoid parabens just in case?
If there’s a potential link between breast cancer and parabens, it seems like it would make sense to avoid them just in case. The problem with this line of thinking becomes more obvious when you realise that the alternatives to parabens have their own disadvantages.
Very few products are safe without preservatives
One way of avoiding paraben exposure is to stop using preservative-containing products altogether. The problem is that cosmetic chemists started putting preservatives into products many decades ago for a very good reason. Every time you open a product it comes into contact with air, which contains microbes like bacteria, yeasts, moulds and fungi that can land and start growing in your product, reaching dangerous levels even if the product still looks and smells fine. If you then apply the contaminated product to your skin, these microbes can cause infections like staph, and can even lead to blindness if they get in your eye. Many microbes also produce allergens and irritants that cause skin reactions.
Unless your product has been manufactured under sterile conditions and remains completely sealed in an airless container when you use it, there’s a good chance that microbial overgrowth will happen if there isn’t an effective preservative system in place. This is especially true of products containing natural ingredients that can act as food for microbes.
Water-free products technically don’t need a preservative, since microbes need water to grow. But as the EOS lip balm black mould controversy of 2015 showed us, water-free products won’t stay water-free if used in wet conditions. Although you can get away with preservative-free waterless products, you probably need preservatives somewhere in your routine.

Alternative preservatives have problems
Another potential solution is to use non-paraben preservatives. While this seems like an obvious and easy fix, there are a whole bunch of problems here too:
- We don’t know much about the health effects of new preservatives: Parabens have been used for almost 100 years by billions of people, and there have been numerous studies done on them. That means we have a pretty good idea of their health effects, which have been minimal so far. On the other hand, newer preservatives haven’t been used for as long and haven’t undergone as much testing, so their health risks (especially long term) are unknown, and could be far worse.
- Alternative preservatives are more problematic or less effective: One of the key reasons why parabens are so popular is because they’re effective at very low concentrations.
- “Natural” preservatives are a lot less effective, so you either have a poorly preserved product with the issues discussed earlier, or you end up with products containing a whole lot of preservative. Since natural preservatives are often irritating and allergenic essential oils and organic acids, many more people will experience reactions with the product. Many natural brands use synthetic preservatives out of necessity, since using natural preservatives only often isn’t feasible.
- Alternative synthetic preservatives are also problematic. Parabens are by far the least irritating and allergenic preservatives on the market, with an allergy incidence of 0.5-1.7% in patch-tested individuals in the US and Europe (it’ll be lower for the general population). For other common preservatives, the incidences reported in this review are:
- Formaldehyde: 9% (US), 2-2.5% (Eur)
- Quaternium-15: 9% (US), 1% (Eur)
- Diazolidinyl urea: 2.7-3.7% (US), 0.5-1.5% (Eur)
- Imidazolidinyl urea: 2% (US), 1% (Eur)
- Methylchloroisothiazolinone/methylisothiazolinone (MCI/MI) 2.3-2.9% (US), 2-2.5% (Eur)
Verdict
Here’s a list of some of the organisations who have stated that current use of parabens is unlikely to be linked to cancer:
- European Commission Scientific Committee on Consumer Safety (SCCS)
- Cosmetic Ingredient Review
- National Cancer Institute
- US Food and Drug Administration
- Cancer Council Australia (also here)
- Centers for Disease Control and Prevention (CDC)
- Health Canada
I won’t be purposely dousing myself in parabens, but I won’t be avoiding parabens either – I feel that it’s far less risky using preservatives that have been tested by scientists and consumers for decades, over brand new ingredients that don’t have anywhere near the same track record. However, I do agree that there are some questions that really need answering with further research.
If you’re still keen on avoiding parabens, I would follow the European Commission’s precautionary approach: methylparaben and ethylparaben are of little concern, propylparaben and butylparaben warrant more caution.
Additional Note
I’m still really annoyed and downright baffled that 13 whole years after the 2004 study, there still haven’t been any published comparisons of paraben concentrations in the breast tissue of women with and without breast cancer, or even a comparison between their urine paraben levels. Comparison with a control is pretty much the first thing you learn about experimental design. I realise that it’s more difficult to biopsy healthy breasts, but surely urine samples aren’t that difficult to collect? A convincing result could change everything. I suspect that there will be no correlation. This 2012 study found higher paraben levels in non-cancerous tissue than was found in tumours in the 2004 study (though in different subjects), and multiple studies have found parabens in pretty much the entire population (98%, 99%, 100%). It’s possible that a study has been done, but publication bias happened, or perhaps the researcher didn’t find what they were looking for…

References
Key Studies
In vitro and rat estrogenic study: EJ Routledge, J Parker, J Odum, J Ashby & JP Sumpter, Some alkyl hydroxy benzoate preservatives (parabens) are estrogenic (full text), Toxicol Appl Pharmacol. 1998, 153, 12-19.
Breast tumour study: PD Darbre, A Aljarrah, WR Miller, NG Coldham, MJ Sauer & GS Pope, Concentrations of parabens in human breast tumours (full text), J Appl Toxicol. 2004, 24, 5-13.
Commentary on the breast tumour study including breast cancer hypothesis: PW Harvey & DJ Everett, Significance of the detection of esters of p-hydroxybenzoic acid (parabens) in human breast tumours (full text), J Appl Toxicol. 2004, 24, 1-4.
Criticism of the 2004 Darbre Study
R Golden & J Gandy, Comment on the publication by Darbre et al. (2004), J Appl Toxicol. 2004, 24, 297-299 (author reply 299-301).
AM Jeffrey & GM Williams, The paper by Darbre et al. (2004) reports the measurement of parabens in 20 human breast tumors, J Appl Toxicol. 2004, 24, 301-303 (author reply 303-304).
C Flower, Observations on the paper by Darbre et al. (2004), J Appl Toxicol. 2004, 24, 304-305 (author reply 305-306).
Critical Reviews of Parabens and Endocrine Disruption
R Golden, J Gandy & G Vollmer, A review of the endocrine activity of parabens and implications for potential risks to human health (full text), Crit Rev Toxicol. 2005, 35, 435-458.
D Sasseville, M Alfalah & JP Lacroix, “Parabenoia” Debunked, or “Who’s Afraid of Parabens?”, Dermatitis 2015, 26, 254-9.
Other Excellent Articles on Parabens
Personal Care Truth: Parabens in Perspective (10 part series)
Science-Based Medicine: Breast cancer myths: No, antiperspirants do not cause breast cancer
Joe Schwarcz’s The Right Chemistry: Paraben phobia is unjustified
Just About Skin: The REAL Truth About Parabens – They’re Nothing to Worry About







Thank you for this! This exact topic was a discussion point on my blog in the comments. I noted that not all parabens are the same (propylparaben and butylparaben seems to be key ones to keep an eye on). And the avoid “just in case” argument didn’t fly with me simply because there just aren’t good alternatives currently to prevent bacteria / moulds etc, which are far more worrisome to me.
I feel like the whole parabens debates aligns with the MSG debate… people only remember an older study that cause widespread panic, but without the scrutiny on the methodology of the studies. Your point about other estrogen-mimicking chemicals is quite interesting – I’m going to read up more on that.
Thanks again! I really appreciate the research you put into your posts. 🙂
Thanks so much! I agree, it’s a lot like the MSG debate, and the GMO debate with the photos of the rats… It’s a lot easier to convince people with emotion than science!
Great post!!
Thank you! 🙂
Thanks for this review, it’s very helpful!
Thanks! I’m glad you liked it 🙂
Thank you so so so much for this post Michelle! I saw your Instagram post and straight away it gave me that feeling of massive relief and happiness because this topic needs to be talked about! – I also wrote a huge essay on your Instagram post because I’m also very passionate about this issue of people perpetuating the idea that parabens are bad. That study where parabens was found in cancer tissue – how does that automatically translate to “parabens caused the growth of cancer tissue”?!
You’re right, it’s a game of whispers and in this age where people want to know in 2 seconds what’s good and what’s bad, that’s what’s made people fall for this. And advocate it strongly as well!
To be fair, it does take a degree / some deep reading into research papers to really understand the discrepancies. For instance, when some studies say that “we conducted this experiment in one cell”, that definitely is not conclusive to it being true when looking at the entire human body as we are more than just one cell.
You have just busted this parabens myth big time!! This post made my day! ?
Thank you so much! It’s definitely difficult to work out what’s happening behind all the marketing, and it’s very frustrating when I see beauty bloggers reference the EWG or other sources unthinkingly because everyone else references them, without critically assessing the content of their arguments. Looking forward to seeing your blog when it launches! 🙂
I would like to hear your opinions about DMDM Hydantoin, a preservative used in lots of cosmetic items, many thanks.
I’ll have a look into it!
This is a great blog post! Please keep it up!
Thank you so much!
Thank you so much for writing this! It is a ray of light in a paraben-bashing world.
Thanks so much! I’m glad you appreciated it 🙂
Thank you Michelle, your blog is heaven for skincare science.
Thank you so much! ^-^
Excellent post!
Any thoughts on methylisothiazolinone are much appreciated!
Love your site.
Very thorough post! A very enjoyable read indeed. I’m glad that you can break down hard science into a easy read for us common folk. I definitely will care even less about parabens now since I’m not sensitive to them anyway. Looking forward to the next science focused topic!
I have been purposely avoiding parabens for nearly two years after being scared out of mind by reading EWG’s site as well as a few other things. This has calmed me down quite a bit about them and has me rethinking how I shop for skincare and makeup items. Thanks so much for this post!
Thanks so much for this post! I’ve seen a number of blogs weigh in on why you don’t need to avoid parabens, but this is by far the best post on parabens I’ve come across. I love that you walk us through the paraben studies and their shortcomings in an unbiased and informative way. This was a thoroughly entertaining read and very easy to understand (well, keeping in mind that I’m a science student so I have a bit more knowledge than average). I’ve just recently found out about your blog and I’m now a big fan. I love that you cover K-beauty products as well! By the way, I’d love too see more posts on the science behind popular K-beauty skincare ingredients (if possible! In my own research I’ve found good studies on many of them to be scant.) Anyway, keep up the awesome work! ?
Thank you so much! I’ve done snail slime but I’ll have to look into more 🙂
I love that you take so much time to dive into the science behind make up! And you actually take the effort to cite all these studies. Sincerely appreciate your work! c:
Thank you so much! 🙂
Have you seen the new study out of the Silent Spring Institute by Dale C Leitman? It seems to say that adding one other ingredient to the paraben increases the harmful affects. Does the concern raised have any merits?
https://ehp.niehs.nih.gov/14-09200/
I’ve seen it, but I’m still really unconvinced without in vivo measurements…
Thanks so much for this post! Great insight, helps with decision making when I buy cosmetics now. Really like how easier you made for regular non-scientific people like me to understand.
Can you also do a post looking into the other Preservatives and how ‘good/bad’ they are? The few you mentioned: Formaldehyde, Quaternium-15, Diazolidinyl urea, Imidazolidinyl urea, Methylchloroisothiazolinone/methylisothiazolinone, phenoxyethanol.
Very often I see products claim they are ‘paraben free’ but have the above ingredients instead. It would be great to see how those affect us.
Thank u for your post michelle… i totally agree with u… but somehow i thought like this, even though the amount of parabens in our skincare and any other products is so small, but since we always use so many make ups, skincares, or so many other parabens contain products everyday in our live and many times a day, is it possible that all that parabens accumulate in our body and because of that it become harmfull to us?
Thank you for this enlightening post — I’ve honestly been concerned reading about parabens, and have noticed the proliferation of a lot of paraben-free products (at a significant price premium, of course) but I wasn’t too clear on what the quantified risks are.
You are my fave blogger (am I using the right term?). It’s so refreshing to read about skincare without the hype to go and buy. Much as I like some beauty editors, I don’t think they very selective about their recommendations and so now I have become a little jaded and suspicious.
Hi Michelle!
Thank you for this overview. I’m with you in that I don’t avoid parabens in products for myself. However, I’m wondering about their safety during pregnancy and lactation. On PubMed there are several recent studies that look into that aspect. Here is one as an example https://pubmed.ncbi.nlm.nih.gov/32283359/
It found correlation between some adverse outcomes and parabens concentration in meconium. Obviously something like that is not conclusive since cohort studies are only the beginning (and yet they’re pretty important in the public health disciplines), but it’s definitely the line that seems to be studied more now (as opposed to the cancer link. Because let’s be real, cancer causation is not the most straightforward thing to prove.)
So I was wondering what your thoughts are on these more recent studies. Are the results even well calculated?
Another concern people have is that when we use many different layers of skincare, our exposure to parabens increases, so even though they are limited per product, in reality we get a higher “dose.” Also would be interested in your thoughts as a scientist on this.
Thank you! Bug fan here 🙂
Oops sorry! I meant big fan, not bug fan…
I am one of the unlucky people that has an allergic response to Parabens.
I was patch tested in 2000 and was initially very relieved when I only reacted to one chemical of the 108 they tested. Unfortunately for me I’m allergic to 4 Amino Benzoci Acid, Para Amino Benzoic Acid and Octyl Dimethyl Paba which in cosmetics is known as Paraben.
I found out that Parabens are everywhere: skin care, hair care, hair dyes, make up, clothes dyes and local anaesthetics. I also have to watch for cross reactions with: folic acids, sulphonamides, AZO dyes and artificial sweeteners.
So while I can understand your annoyance with the 2004 study, as someone who has contact dermatitis caused by Parabens I am very grateful for it. I now don’t have to have arguments with people to obtain chemical contents lists for makeup and there are more ‘Paraben-free’ products that I can actually use as companies are at least attempting to look at using other preservatives besides Parabens.
I guess you’re one of the lucky beneficiaries of the myth then 🙂 A lot of coeliac sufferers have also mentioned that the “gluten-free” trend was fantastic for them, since there were so many more foods they could suddenly eat, even though it’s based on BS!
I know this is an old post but is the noise around the environmental toxicity of parables also fear mongering along the lines of reef safe sunscreens or is there legitimate concern?
it seems like scss already gave their okay for propylparaben? (mar ’21) https://ec.europa.eu/health/sites/default/files/scientific_committees/consumer_safety/docs/sccs_o_243.pdf