
Are chemical or physical sunscreens better? I touched on this in my Sunscreen and Make-up video, but a lot of people have been asking me to talk more about it, so I’ve expanded on the topic in this post, which comes in video form as well!
Click here for the video – scroll down for the blog post “summary” version with references and product recommendations (which is still somehow 1500+ words long…).
What Are Chemical and Physical Sunscreens?
The active ingredients in sunscreens are often divided into two categories:
- Physical sunscreen ingredients (more correctly known as inorganic sunscreen ingredients) are zinc oxide and titanium dioxide.
- Chemical sunscreen ingredients (more correctly known as organic sunscreen ingredients) are everything else.
You can have sunscreens containing only organic filters, only inorganic filters, or a combination of both.
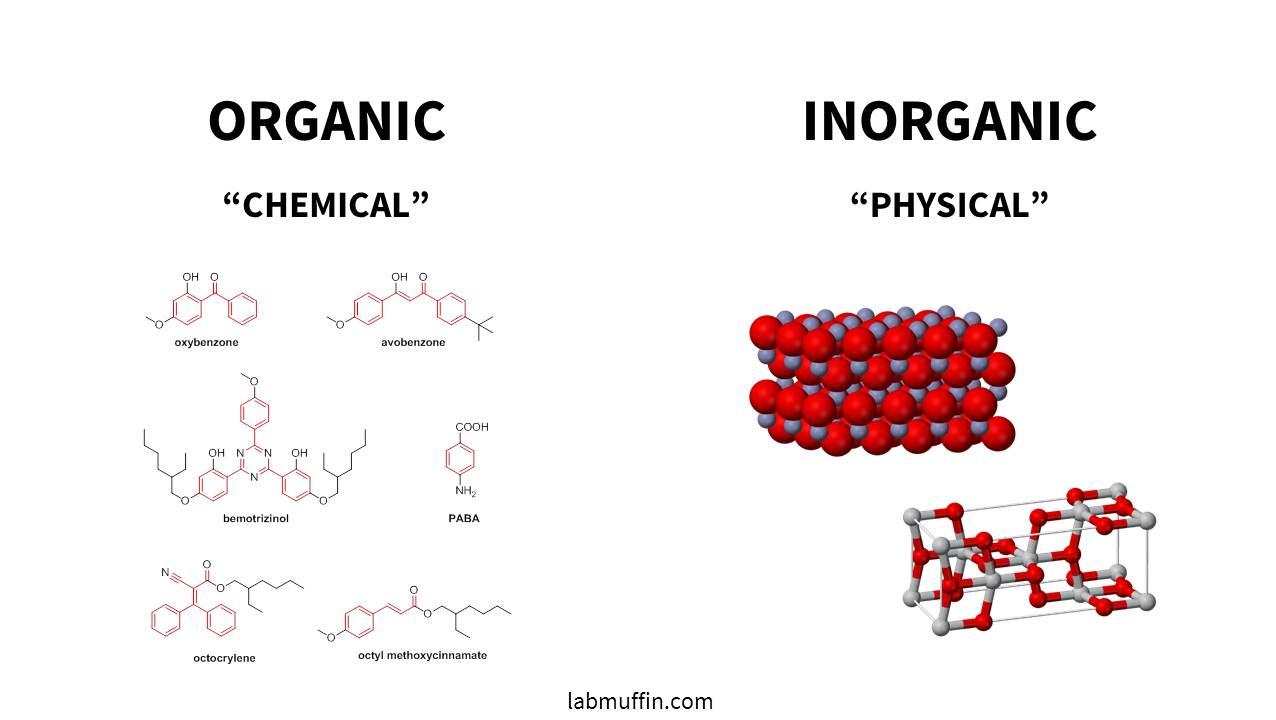
The reason organic (carbon-based) and inorganic (not carbon-based) is a better classification than chemical/physical is that there’s overlap between how they work. Both types work by absorbing UV and turning it into heat. Inorganic sunscreens also scatter and reflect about 5-10% of the incoming UV, as do some particulate organic sunscreens like Tinosorb M, so really they should be classified as both chemical and physical.
Differences Between Chemical and Physical Sunscreens
The big differences between them that you should consider are:
Protection Level
SPF 50+ is pretty common with both types of sunscreen, but broad spectrum protection (that includes protection against longer wavelengths of UVA) is where there’s a difference.
Organic sunscreens give higher, photostable protection from UVA if you use newer filters like Tinosorbs S and M, and Uvinul A Plus (not yet available in the US). The more common avobenzone gives really high UVA protection, but it breaks down in UV so you have to be diligent about reapplication (although some formulas stabilise avobenzone so it breaks down slower, and you should diligently reapply sunscreen anyway if you’re spending a lot of time in the sun).
You can’t really tell what the UVAPF of a sunscreen is without actually testing it (sunscreen calculators like the BASF Sunscreen Simulator don’t accurately predict the protection of a finished formulation and should be used for formulating purposes only). Very roughly speaking, for sunscreens with a tested, publicly available UVAPF value, inorganic sunscreens get around UVAPF 20 max, while all the highest UVAPF sunscreens are organic (e.g. Bioderma and La Roche-Posay can get around UVAPF 40 – if you’ve seen higher, let me know!).
White Cast
Organic sunscreens tend to leave less white cast while inorganic sunscreens tend to be very white (especially if they have titanium dioxide).
I recently got rematched with MAC shades and it turns out my skin has gotten paler thanks to my religious sunscreen use – I’m now closer to NC20 than NC25. But even though I’m on the paler end of the spectrum, inorganic sunscreens still commonly make me look mime-like if I apply 2 mg per square centimetre to get full protection, which translates to about ⅔ of a quarter teaspoon for me. This makes it hard to apply a generous amount, and since SPF scales with how much sunscreen you apply, this is an issue.
Related post: Video: How Much Sunscreen Do You Need For Your Face?
Exhibit A:

Texture
Sunscreens often have really thick, greasy textures and depending on your skin, one type might suit you better. I personally prefer organic sunscreens, since I’ve found quite a few that feel like moisturiser.
Sensitive Skin
If you’re in the US, avobenzone is in almost all broad spectrum organic sunscreens (except for the ones containing L’Oreal’s patented Mexoryl SX). Avobenzone is a common irritant and allergen, so it tends to be unsuitable for sensitive skin, and you’re left with inorganic sunscreens only.
If you’re elsewhere in the world, the newer UVA1 filters aren’t particularly irritating or allergenic.
A few other organic sunscreens also tend to cause allergic and irritant reactions: octocrylene, oxybenzone, avobenzone, PABA, Padimate O and enzacamene.
Myths About Chemical and Physical Sunscreen Differences
There are a whole bunch of myths surrounding the topic of chemical and physical sunscreen. Kind of Stephen has written a great article on most of these myths where he goes into more detail about the studies.
You need to apply chemical sunscreens first and physical sunscreens last
You need to wait for chemical sunscreens to work but physical sunscreens work immediately
Both of these myths are based on the myth that chemical sunscreens need to absorb into your skin and bind before they work – but both types of sunscreen work straight out of the bottle.
All sunscreens just need to form a continuous film on your skin, and they’ll work, so you can apply them exactly the same way.
Physical sunscreens are better because they’re natural
Natural things aren’t better than synthetic, man-made things – I have a whole video on this topic: Video: Are Natural Beauty Products Better?
Even if they were – physical sunscreens aren’t even natural. They’re processed to get rid of toxic contaminants, and often need to be coated in synthetic chemicals to stop them from being photocatalytic, and prevent them from clumping up and causing patchy protection.
Chemical sunscreens produce heat which is bad for your skin
The amount of heat produced from UV by sunscreen is really, imperceptibly tiny. There’s also only a 5% difference in the heat produced by the two types of sunscreens, since physical sunscreens also absorb about 95% of the UV they protect you from.
You need to reapply chemical sunscreens if they’re exposed to sun, but not physical sunscreens (sort of true)
This myth is based on the idea that chemical sunscreens aren’t photostable, which means the molecules break down after absorbing too much UV and need to be replaced. But these days a lot of chemical sunscreens are photostable. The most photounstable combination is avobenzone and octinoxate, so it’s a good idea to reapply sunscreens with that combination frequently.
But you should really be reapplying ALL sunscreens, even without sun exposure. The main reason why you need to reapply sunscreen is that sunscreen shifts around and off your skin throughout the day, esepcially if you’re active.
However, studies on daily sunscreen use found significant benefits even with once a day application and with regular activity.
You can use less of a physical sunscreen (sort of true)
You need to use the same weight of chemical and physical sunscreen. But physical sunscreens tend to be denser, so on average you can use around 20% (⅕) less by volume.
Chemical sunscreens are hormonal disruptors (sort of true)
This is a complex topic.
There are a LOT of different chemical sunscreens. They have:
- different absorption rates – many new sunscreens don’t absorb into your body at all
- different UV efficiencies – so different amounts of each ingredient needs to be used to get the same UV protection, so our exposure to them differs
- different effects on the body – some have no known effects, some have hormonal effects
So you can’t make any blanket statements.
A lot of the fear is around finding these ingredients in breast milk and in urine. This sounds scary, but:
- detection methods are so sensitive now that teeny tiny amounts can detected, so finding something doesn’t mean there’s enough of it to have any effect
- the ingredient has to actually do something to be worth worrying about – for example, water is in breast milk and urine too
Out of the sunscreens, the one with the strongest hormonal effect is oxybenzone, according to in vitro studies on cells and animal studies (there haven’t been any human studies that have found a hormonal effect at the time of writing). One dermatology paper calculated that in humans, you’d need to use oxybenzone sunscreen continuously for 277 years to get the equivalent amount to cause a noticeable hormonal effect, so it’s considered safe.
Other sunscreens that have had hormonal effects in studies are enzacamene, padimate O, octinoxate and homosalate, although their effects are several orders of magnitude lower than oxybenzone, so the amounts you’d need would be even bigger.
Physical sunscreen nanoparticles are bad for you
Zinc oxide and titanium dioxide nanoparticles could potentially make reactions happen in your body, particularly in UV light. But the studies so far have found that the nanoparticles don’t get very far into the skin (only to the dead layers of the stratum corneum). It’s possible that nanoparticles will penetrate further if you apply them on broken skin, but they’re currently considered safe.
Sunscreens turn into free radicals
Both chemical and physical sunscreens have been found to react in UV light to produce highly reactive free radicals which can cause damage to surrounding substances, much like UVA. But these studies were generally performed in vitro with cell lines, while clinical studies on humans have found that sunscreens will reduce skin cancer and aging.
These reactions also only happen when enough UV hits the sunscreen, so they will generally only occur in the sunscreen film or in the upper, dead layers of your skin (same deal with retinyl palmitate).
Bottom Line
So at the end of all that: use whatever sunscreen works best with your budget, skin type (oily, dry) and skin concerns (fading hyperpigmentation, sensitivity, allergies, acne). Ideally, it should have high protection (SPF 30+, broad spectrum).
I hope this answered a lot of people’s questions about the different sunscreen ingredients! If you want to learn more about sunscreens, there are a whole bunch more posts I have on them – here are a few:
- How Much Sunscreen Do You Need for Your Face? (Video)
- How to Get Vitamin D and Stay Sun-Safe (Video)
- Sunscreen FAQs (Video)
- What Does SPF Mean?
- Multi-Sunscreening
Some of these products were provided for editorial consideration, which did not affect my opinion. This post also contains affiliate links – if you decide to click through and support Lab Muffin financially (at no extra cost to you), thank you! For more information, see Disclosure Policy.






This is article is so helpful. Thank you for breaking down these myths I didn’t even know that people believe some of these myths. I’m saving this blog post for future use in case I ever run into someone who mistakenly believes some of these misconceptions about sunscreen.
Hi, thanks for the great overview. I’d love to see your thoughts on reef safe sunscreens as well. I know oxybenzones are particularly bad for corals, but it’s really hard to find guidance on good sunscreens, especially for those of us with darker skin and risk of white cast!
It’s a good idea to avoid oxybenzone, octinoxate and nanoparticle inorganic sunscreens if you’re going into the water!
We all shower and bathe, then the water goes “downhill” eventually into the ocean, doesn’t it? Shouldn’t we all avoid oxybenzone, octinoxate, and several others in order to avoid bleaching and killing coral reefs? I am so confused!
When things go down the drain, they get diluted to the point where they’re only present in very very small quantities – much smaller than the amounts used in the coral studies. It’s like how all food contains mercury, but the amounts are so small they won’t interact with the body to any large extent.
Hi Michelle,
Love your website and all the information you share.
Have you heard of sunscreen soaps (e.g. Klenskin brand)? If so, what do you think about them? I couldn’t find much information on them and I have family members wanting to try it.
Thank you!
I haven’t tried them – I’m skeptical, but would love to see their testing methodology!
I absolutely LOVE LOVE LOVE your blog!
What do you thank About Paula’s Choice sunscreens?
Unfortunately I haven’t tried many of them, but they’re formulated for the US market so no newer filters 🙁
I always say mineral vs. synthetic.
Hello there. what is the best sunscreen for acne prone skin, especially the cystic hormonal kind. thanks, enjoyed the article!
I am very happy that in the EU we have access to the new formulas with ease, it helps me keep myself and the kids protected from UVA and UVB.
Thank you Michelle for another greatly informative and easily understood video post. You and your work are very much appreciated!
I do have a question on the texture of most facial sunscreens. Why are the majority of them in such a greasy base? Is it because of the size of the particles or that it needs to stay on the surface of your skin or ……what? Makes it difficult to find a workable sunscreen to use even without makeup. The best that I’ve found, so far, for my combination skin is LaRoche-Posay 50 Mineral Ultra Light Sunscreen Fluid and SkinCeuticals Physical Fusion UV Defense SPF 50 Universal Tint. They have almost the exact ingredients list and dry down enough in the recommended eight minutes to apply makeup. They are pricey though :/
Thank you!
I think it’s mostly to form a film that stays intact on your skin, and for water resistance. Physical sunscreens also need to keep the particles from clumping up, and a thick base basically makes it harder for the particles to move around and find each other.
Ahh, I see. I just kept wondering why the greasiness every time I sigh and nix yet another bottle of facial sunscreen. The textures just seemed counter to the goal of getting people to wear the recommended amount of sunscreen. Well maybe science will eventually produce one that will be easier to use as recommended 😉
Thank you, thank you for answering! You and your brain are awesome!
Hello and thank you for the post.
There is this UVA65 sunscreen:
https://www.uriage.com/LB/en/products/bariesun-xp-creme-spf50-1
It is a joke concerning its cosmetic elegance. By far the worst I have every put on my skin.
I think the Nivea Sun Protect Plus UV Milky Essence SPF 50+ has been discontinued.
Keep up your work!
Ugh that’s unfortunate! The only Uriage sunscreen I tried a few years back balled up on my skin really badly.
Dear Lab Muffin,
I am a big fan of your blog, please keep the posts coming! <3
I would love to use more of "chemical" sunscreens as some of them feel really nice on the skin, but my problem with them: no matter how far from the eyes I apply it, not higher than the cheekbones, the sunscreen eventually gets into my eyes sometime during the day (I suspect with the eyelashes' movement), and it STINGS the hell out of it. My eyes go read and watery by the end of the day which shouldn't bee too good for them either. I do use a mineral powder on top of the sunscreen to set it better, but it happens all the same. None if this happen with physical sunscreens (but I hate the feel of even the best of them).
I wonder if you experience any eyes irritation because of the sunscreens? Maybe there is one that doesn't sting the eyes? (I am in Australia as well). Or maybe there is a secret how to apply it better to avoid eyes irritation?
Thank you for your work again:)
I’ve been purposely trying out new sunscreens on my eyes and eyelids lately – I’ve found that the stinging for me is usually from alcohol, but if it happens later I would guess that it’s either avobenzone or octinoxate. Maybe try some of the newer sunscreen ingredients? I’ve found that Mecca To Save Face SPF 50 doesn’t sting my eyes, and Mecca are happy to give out a sample in a pot to try before you invest 🙂
Thank you Michelle! I’ll try Mecca’s sunscreen:)
Hello Michelle
Thank you for wonderful overview.
I am under impression that chemical sunscreens are more reliable in general.
I have been suggested Dermalogica Oil Free Matte SPF 30+ for oily skin. What’s your thoughts about that, please.
Best Wishes,
KK1968
I have that feeling too – partly because chemical sunscreens sink into skin a bit more so they shift around less, although there’s the photoinstability issue with older sunscreens.
I haven’t tried any Dermalogica sunscreens before unfortunately!
Hi Michelle, I would like to know what you think of Biore sunscreens
thanks.
I haven’t used them in a while, but I really liked the 2015 Aqua Rich Watery Essence, although I felt that it moved around a lot on my skin.
I’m using actinica sunscreen which has PPD 39 I think. I read somewhere on reddit, according to a french blogger, that it’s not photostable after 2 hours, uva drops to 46%. Idk, I have to order it overseas as it stop selling in Australia. I have another sunscreen skinstitut age defence, really good sun filters tinorsorbs, univual, no Avobenzone. It has same dewy finish as actinica.
I use moogoo buttercup SPF 15 sunscreen which has 19% zinc oxide around my eyes, it’s good no stinging or watery eyes. Their SPF 40 version is way too thick. SPF 15 with 19% zinc oxide is really good but no one buys the SPF 15 due to the 15 which is a shame. I then apply chemical sunscreen to rest of my face. It’s the only way I can get chemical sunscreen not to irritate my eyes. I like buying tinosorb sunscreens for the UVA protection. On my body though I use Cancer Council Active Dry Touch sunscreen- high 5% Avobenzone sunscreen- literally the most invisible weightless feel sunscreen I tried. Bit too matte for my dry face though.
So glad I found your blog. Thank you for all the info on one of the most confusing and myth-surrounded topics, sunscreen. It seems so many skincare enthusiasts are still looking for their HG, which offers reasonable photostable protection while still being cosmetically elegant. Haven’t found mine yet ? but will keep looking. Looking forward to your next posts.
Thank you for this Michelle, you’ve blown my tiny mind.
I have quite sensitive skin so have been using inorganic sunscreens for the last 10 years (every day) because most organic sunscreens I tried made my skin feel like it was burning and/or come out in a rash. Recently, I got some freckles lasered and despite my everyday SPF 50 application, the pigmentation returned, grrr.
But watching this prompted me to go out and find some high PPD/UVA sunscreens. The new filters don’t irritate my skin at all, which is great, but more importantly after a few weeks I noticed that my pigmentation had really faded!
This was excellent proof of the superior PPD/UVA of the new filters, I’m totally converted.
Generally agree with you on the shininess of the French sunscreens, though the La Roche Posay Anthelios Anti-Shine is good for my combination skin. Just about to try the Avene emulsion. The tinted Bioderma is quite good too, though has a lower PPD.
Some PPDs I’ve been able to find:
La Roche Posay Anthelios Dry Touch tinted- 22 haven’t tried, PPD to low to bother
LRP Anthelios Dry touch – 31 not shiny, a little sticky, could make a good primer once it dries
LRP Uvidea tinted – 34 shiny, decent coverage, breaks up though
LRP Anthelios B.B. – 40 for dry skin, haven’t tried
LRP Anthelios Tinted Fluid – 34 shiny, very strong scent
Bioderma B.B./Aquafluide tint – 26 a little shiny, slight coverage, light tint a bit dark for my NW15 skin
Bioderma creme teintee – 37 not available in Australia 🙁
Avène emulsion – 38
I’m so impressed I actually was motivated enough to type all this out on my phone.
Thanks so much for all that info! I wish it was easier to find all these values in Australia!
Do you have any recommendations for waterproof sunscreens for the body and face?
This is a great article. Thank you!
Just one thing I’m wondering if you could clarify.
It’s mentioned that inorganic reflects UVB (the sunburning component of UV) but I can’t seem to work out if it also reflects UVA. In the study referenced, they mention “protection from sunburn” (UVB).
So does it reflect about 5% of UVB and absorb the remainder? Does it reflect the entire UVA?
Thanks!
There’s basically no reflection below 370 nm.
Wow, such a great article for sunscreen lovers. Answered so many of the concerns . Thank you !
Hi Michelle!
Thanks for the comprehensive post!
I’ve recently tried to wrap my head around the science behind sunscreen recommendations for melasma. I’ve always believed that inorganic (high zinc) sunscreens were the way to go. From reading this, I take that a high PPD and adequate protection against visible light really should suffice, regardless sunscreen category. Or is there something more to melasma? I’d really I’d love to hear your take on this!
(I’m in Europe and have the new filters readily available, yet order zinc sunscreens from the U.S… The irony!)
Yep, high PPD is what you need! There’s also the higher incidence of allergies, but if you’re not allergic then it shouldn’t be an issue.
THANK YOU SO MUCH! I’ve been going raving mad with fear over these past few months especially with everyone calling out chemical sunscreen. I literally looked through my sunscreen stash (I live in the Middle East and bulk buy for myself and the family on sales because “Middle East prices ¯\_(ツ)_/¯”), checked the ingredients on all my beloved ACM, Eucerin, LRP, Nuxe, Bioderma and Nuxe sunscreens, which are of course all chemical and all extremely expensive here, and had just made up my mind that I’ll use up this lot, risk dying or whatever and buy my zinc based ones next year and go around looking like a brown ghost of Christmas past from then on.
Thank goodness I don’t have to do that anymore and can go on slathering myself in my Goop unapproved chemical sunscreens.
Have you tried wearing Zinc in 50 C weather? It is a losing war in which no brown girl comes out looking good.
sources for not needing to apply sunscreen 15 min prior?
Check out the video – there’s footage of the sunscreen absorbing UV right after it’s applied to white paper.
I appreciate how you said that sunscreen will reduce skin cancer and aging. I am scared of getting something like skin cancer. It can happen to anyone so I’ll be sure to get as much sun screen as possible.
I find that your article did not fully understand the article you had quoted that physical sunscreen and chemical sunscreen function “the same”. Technically yes, they DO function the same. But realistically, they DO NOT. It is because physical sunscreen while they do absorb UVB, the absorption is SO LOW, (5%) as quoted by the article, that the effectiveness of mineral sunscreen absorbing UVB and providing SPF is ridiculously low to be touted as having the SAME FUNCTION as chemical sunscreen. As a PHD yourself, I do hope that you can create informative science for layman without misconstruing science. In fact, to have the best SPF, combination of mineral sunscreen & chemical sunscreen is important to protect from UVA & UVB. To say that mineral sunscreen function THE SAME as chemical sunscreen insinuate and misleading information that consumer would feel MINERAL SUNSCREEN IS SUFFICENT FOR SPF — which is immensely untruthful.
The article says the absorption is 95%, reflection and scattering is 5%. The important thing when looking at sunscreen is the final tested SPF, regardless of whether it’s chemical or mineral.
Hi Michelle! I found your blog really helpful. This is the best blog I have found till now regarding sunscreen. Before I read your blog I did not know that we even have organic(chemical sunscreen) and inorganic( physical) sunscreen. I just used to buy random sunscreen and apply to my face and body. As I have sensitive skin, my skin is easily irritated and breaking out. I was really insecure about my skin. Through your blog I found out I was using physical sunscreen that did not suit my skin . Thanks to you that from today I will shift to chemical sunscreen. Please post more blog related to skin care product.
Hi, omg I love this website so much! Can u please star to post a “work cited” after your articles with the info about where ur research is from? I hate to sound like a teacher but I’ll be making a college research on this and I need to put all my references. (also I’ll definitely be citing your work bc its incredible!)
The research is usually linked, if it isn’t it’s probably linked in the video or in a past article 🙂