Vitamin C is an amazing skincare active, but ascorbic acid can be annoying in products. That’s why vitamin C derivatives were developed – these are slightly altered versions of ascorbic acid that can get around these issues (irritation, instability, stained fingers, fake tan smell).
Two of the INCI names for derivatives that you might’ve seen on ingredient lists are ascorbyl tetraisopalmitate (often called ATIP or VC-IP) and tetrahexyldecyl ascorbate (THDA or THDC). After a lot of digging, I’m pretty sure they’re the same molecule – or at least, the vast majority of ingredients called ATIP and THDA used in cosmetics are the same molecule.
There are three possible candidates here, which I’m going to call A, B and C:
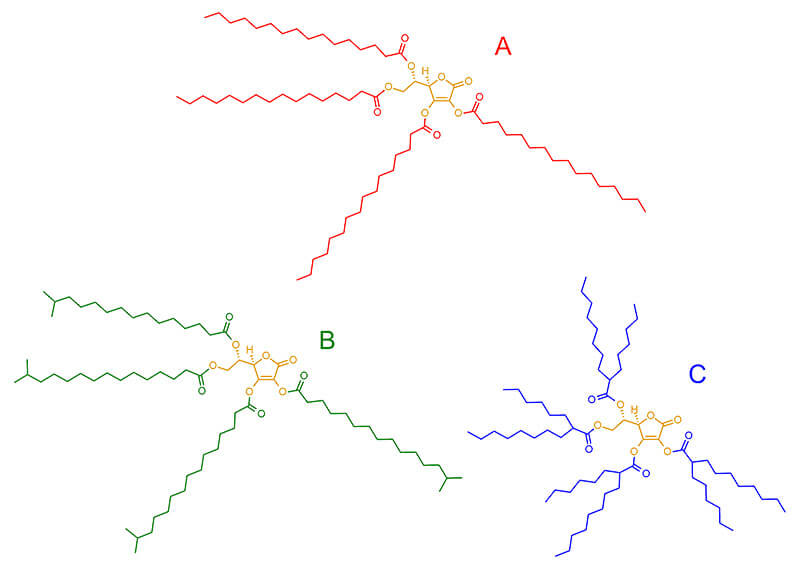
All three have an ascorbic acid core (orange) and 4 of the same fatty acid connected to ascorbic acid through ester linkages.
- A has straight chains of palmitic acid (red), also known as hexadecanoic acid
- B has branched chains of isopalmitic acid (green), an isomer of palmitic acid (isomer means same atoms, arranged differently) that can also be called isohexadecanoic acid
- C has branched chains of 2-hexyldecanoic acid (blue), which is another an isomer of palmitic acid
(You can already see why the names are going to get mixed up…)
Because the three different fatty acids are isomers each containing 16 carbon atoms, the three final structures are also isomers. This means they have the same molecular weight. They’d also be expected to have pretty similar properties, although you’d expect A and B to have slightly higher melting points than C due to the shape of the fatty acid chains.
Which is ascorbyl tetraisopalmitate?
Ascorbyl tetraisopalmitate was the first version developed and mainly marketed by the Japanese company Nikkol Group.
Because this whole thing wasn’t confusing enough, the original 1993 patent from Nikkol is in Japanese, including the chemical names. It mentions isopalmitic acid and tetraisopalmitate, which would seem to indicate Structure B.

But “iso” seems to be used in Japanese as shorthand for “isomer of”, such as in this unrelated patent – which opens it back up to structure B or C.

But a 2009 paper with an author from Nikkol calls VC-IP “2,3,5,6-O-tetra-2′-hexyldecanoyl-L-ascorbic acid” (very clearly Structure C), as well as “vitamin C-isopalmityl tetraester” (which is Structure B if you take the strict meaning of “isopalmityl”, but could also be C if you assume “iso” just means isomer).
They’ve included a diagram of the structure, which is Structure C:
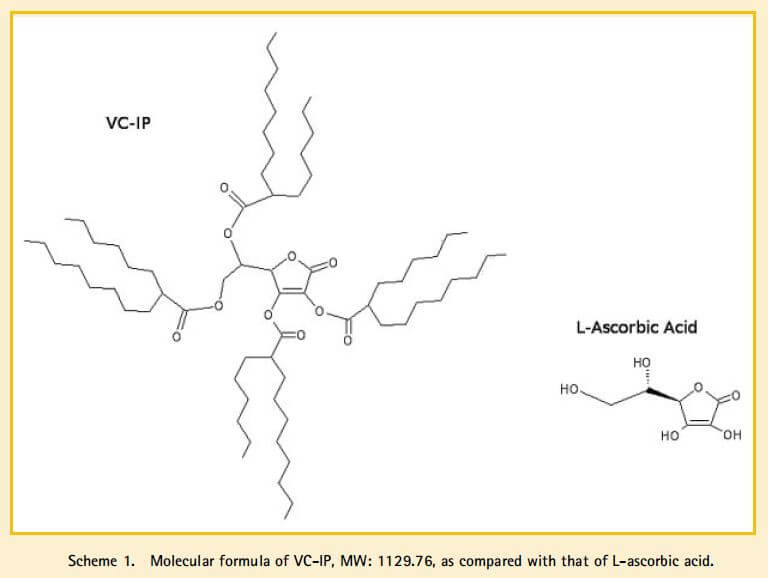
They describe the synthesis using 2-hexyldecanoyl chloride, but the spectral data (1H and 13C NMR, IR) match the 1993 patent, indicating that they’re the same molecule.
So it seems pretty settled that VC-IP, as supplied by Nikkol, is Structure C.
Additionally Greaf, a supplier of ascorbyl tetraisopalmitate explicitly say it’s also known as ascorbyl tetra-2-hexyldecanoate (Structure C). ATIP is also listed for import to Australia via NICNAS with the chemical name 2,3,5,6-tetrakis(2-hexyldecanoate) L-ascorbic acid (Structure C). Additionally, Structure C is listed in Japan as a quasi-drug with the name “ascorbyl tetra(2-hexyldecanoate)”, but is also given a Japanese synonym that equates to tetraisopalmitate ascorbic acid.
Which is tetrahexyldecyl ascorbate?
The name tetrahexyldecyl ascorbate seems to have been popularised by a company called Barnet Products, who are currently listed as one of Nikkol’s US distributors. They sell THDA under the trade name BV-OSC, and their dossier shows Structure C and a scheme for its production from “isopalmitic acid”. Structure C also makes sense, given the naming conventions for organic molecules (“hexyldecyl” would indicate that’s made from hexyldecanoic acid).
There’s been a general vibe that THDA is considered the cheap knockoff of ATIP, but it seems like they’ve been the same molecule all along, and possibly even manufactured by the same company, Nikkol!
Just to really, really confuse things even more, in January 2022 the ingredient supplier Sytheon released a statement saying that tetrahexyldecyl ascorbate (which they sell as VitaSynol C) and ascorbyl tetraisopalmitate were different substances, with different chemical catalogue (CAS) numbers. They clarified that the name THDA would match Structure C while ATIP would match Structure B.
This clarification was done in collaboration with the Personal Care Product Council (PCPC) and the Chemical Abstract Services (CAS # provider), who are pretty big organisations in the cosmetic chemistry/chemistry world.
But it’s also pretty clear from the papers and patents discussed above that the ingredient sold as ATIP has also been Structure C all along – it seems to just have been a translation issue. That means Structures A and B don’t seen to have ever been sold or used in cosmetics, and everything seems to just be Structure C.
Which name is correct?
Neither name is really that accurate – I think the most accurate, reasonably catchy name would be ascorbyl tetrahexyldecanoate.
- Esters are named with the name of the alcohol first – in this case the ascorbic acid’s alcohol groups are being used = ascorbyl
- The fatty acid has a 10 carbon chain (decanoic acid) with a 6 carbon branching off the second carbon (2-hexyl, but numbers are often stripped from INCI names because no one wants to pause to say a number) = hexyldecanoic acid
- The back half of the acid name turns into “-oate” after esterification = hexyldecanoate
- There are four of these = tetra
But ascorbyl tetrahexyldecanoate isn’t listed as an official INCI name while the other two names are, so… bad luck, me.
Hopefully this has made things a bit clearer for those of you who were also agonising over this (possibly a whole three people!).
(Thanks to Stephen for being my sounding board on this topic!)
References
Xiao L, Kentaro Kaneyasu, Yasukazu Saitoh, Terashima Y, Yasunori Kowata, Miwa N. Cytoprotective effects of the lipoidic-liquiform pro-vitamin C tetra-isopalmitoyl-ascorbate (VC-IP) against ultraviolet-A ray-induced injuries in human skin cells together with collagen retention, MMP inhibition and p53 gene repression. Journal of Cellular Biochemistry. 2009;106(4):589-598. doi:10.1002/jcb.22032
Behrens M. Sytheon Corrects Identity of Vitamin C Tetra Esters, Found Skin Benefits. Cosmetics & Toiletries. Published January 11, 2022. Accessed April 14, 2022. https://www.cosmeticsandtoiletries.com/research/literature-data/news/21992979/sytheon-ltd-sytheon-corrects-identity-of-vitamin-c-tetra-esters-found-skin-benefits






Yaaasssss, thank you! I was one of the three people agonising over this but I have only MSc in Biomedicine so this was too much chemistry and too little effort from me to figure it out. I also read the Sytheon statement and thought ok then, they have chemistry department and big regulatory people behind that statement. But wow, you are thorough 👏👏👏.
are we still going to get that part 2 on your vitamin C guide with derivatives?? 🙂
Hopefully! I’m working on it, I posted this so I wouldn’t have to explain it in the video 🙂
Hello yessss I would like to have it as well : I wanted to use vitamin C and I used the C Glow of Geek&Gorgeous…. But after a while I developed irritations on my cheeks 😭😭. My cheeks were kind of red / inflamed … I tried the vitamin C 10% of Vichy (the ampoules) but same problem…
So I would really like to know which derivative is the best for collagen production 🙏
Regarding the antioxidants properties I assume (hope) that a product like the survival 0 of Niod can do the job. But I’m struggling about the collagen part.
I have no knowledge of chemistry and have no idea what any of this means but I love it so much!! <3 <3 <3
First time poster here, (I’ve lurked for a while…). Thank you for the detailed breakdown of the chemical composition! I’ve used traditional Vitamin C serums with L’Ascorbic acid for quite some time but as I’ve gotten ‘wiser’ in years, I have switched to tetrahexyldecyl ascorbate instead. It’s more agreeable with my post-menopausal, sensitive skin.
Love your content!
Which product are you using ?
I admit it has not made me any wiser – I was certainly not one of the three people agonising over it.
But thank you none the less for the effort of educating us on things we didn’t even know were wrong!
Ascorbyl Tetraisopalmitate CAS: 183476-82-6
Tetrahexyldecyl Ascorbate CAS: 183476-82-6
Cosing database.
They’re in the process of changing it – see the article linked in the Syntheon part.
Thank you, thank you!
I have been driving myself crazy over the past few months since I realized that one of the post menopausal skincare changes I needed to make was to switch from ascorbic acid a few times a week to using a derivative instead. It didn’t take long to run across this discrepancy as I tried to learn about the available vitamin c derivatives.
Michelle, thank you for taking this issue up! I guess I would be the third person in this group (I recently chimed in on the discussion over at RealizeBeauty which you undoubtedly followed).
What I found really striking was the fact that a recent review article by Enescu et al. (https://doi.org/10.1111/jocd.14465) misleadingly treats both as different ingredients without giving any source for it (and, in fact, despite citing the 2009 paper as well as other research done on THDA which reference it). I wrote to the corresponding author to bring this to their attention, but so far have not heard back from them.
Glad you appreciate it! To be honest I don’t really think it’s a very high quality review. The lead author who did most of the work seems to be a medical student with a BSc (majoring in biochem and molecular biology, which aren’t relevant to small molecule pharmacology) and the other three authors are MDs who don’t seem to have any solid chemistry or pharmacology background. There’s a lot of incorrect chemistry in it and weird choices of citations. I started doing a thread of the issues on Twitter but I got only got up to Part 2 and I wasn’t really exhaustive…
Thank you for bringing this to wider attention! I guess that makes me a member of the exclusive group of three then…
I joined our little club because a recent review paper on vitamin C derivatives in the Journal of Cosmetic Dermatology (doi: 10.1111/jocd.14465) treats both as separate ingredients, with THDA purportedly being less well-studied. They insinuate that THDA (!) corresponds to either structure A or B without giving any source for that claim, but citing research on THDA that clearly references the earlier studies on VC-IP including the 2009 paper you mentioned…
Rather sad to see that myths and ideas that are discussed somewhere on the internet can make it into a peer-reviewed journal without anyone noticing. I wrote to the corresponding author, but so far have not heard back from them.
Michelle, thank you so much for clearing this up!!!!!!
I have been working on a THDA serum for a while and when finally I had a nice product, our supplier informed me that they have miss labelled all the samples they had previously sent me and the true chemical was ATIP. Then I went through a nightmare trying to find out what was going on. It seemed that this was not the only supplier with unclear information. At least now I have better understanding. I am so glad you did this article!
I am one of three people! It’s been so confusing but now I understand! Thank you so much!!
Late to the party, but this is very helpful! I have lately been using GoPure vit C and liking it. I switched from a different THD serum bc the price doubled (Ghost Democracy). My skin seems to tolerate THD much better than AA products. I just got an email from GoPure saying they have reformulated their serum to contain ATIP instead of THD. I was wondering what the difference between to two was and concerned bc I really liked the THD product. Thank you for posting this.
So thankful for the information you provide Michelle!!
Anna