
We all know what a moisturiser does (it moisturises, duh!) – but what exactly goes in one, and how does it work?
Skin dryness is, quite simply, when your skin loses too much water (the technical term is transepidermal water loss, or TEWL). Moisturisers are used to slow water evaporating, and restore the original soft texture and look of your skin. There are three basic types of ingredients commonly found in moisturisers:
Occlusives
Occlusives slow down evaporation of water from skin by forming a thin, oily layer over the skin (remember from the face washing guide how oils repel water?). These work best when applied to damp skin, such as after a shower. Sometimes these feel too greasy to be comfortable.
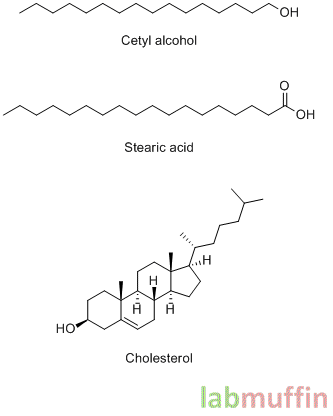
Examples: petroleum jelly/Vaseline (super effective, 98% reduction in water loss in some tests!), lanolin, mineral oil, silicones (e.g. dimethicone), vegetable oils, cetyl alcohol, stearyl alcohol, stearic acid, lecithin, cholesterol, candelilla wax, carnauba wax, beeswax
Here are the chemical structures of some occlusives:
As you can see, they have long zigzags without letters on them. This is important because each corner on a structure represents a carbon atom with a bunch of hydrogens. When you have these long zigzag carbon chains, the chemical tends to be “oily” and won’t dissolve in water well, and won’t evaporate easily, which is why they make good occlusives.
Humectants
Humectants grab onto water from the surrounding environment and draw it to your skin, and keep it there. It also draws some water from deeper layers of your skin (dermis) into the surface layers (epidermis), which can be counterproductive as it’ll speed up water evaporation into the environment. This is why humectants are almost always combined with an occlusive.
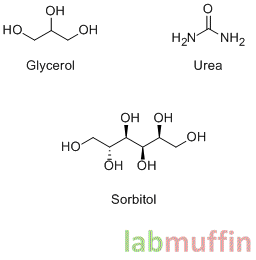
Examples: glycerol, urea, hyaluronic acid, ethylene glycol, propylene glycol, sorbitol, pathenol, honey
These are the structures of some humectants. As you can see, they have lots of OH or NH2 groups attached – these are very good at grabbing onto water.
In our lab, it’s very annoying to make things with lots of OH or NH2 groups, since it’s depressing watching your lovely white crystals turn into sludge before your eyes on a humid day!
Emollients
Emollients soften skin, making it more flexible and smooth. They act by filling in gaps in the surface skin, and assisting in skin repair. They won’t necessarily affect the moisture content of skin and therefore aren’t “true” moisturisers, but since they are usually lipids and oils, and some emollients are also occlusive.
Examples: cyclomethicone, dimethicone, isopropyl palmitate, isopropyl isostearate, castor oil, glyceryl stearate, jojoba oil, propylene glycol, vitamin E, cholesterol
Generally in a moisturiser, you’ll want some of each type of ingredient, but the relative amount will vary according to skin type and climate, e.g. occlusives might not be necessary for someone with oily skin, and may in fact result in pimples, whereas some humectants can be too irritating for people with dry or sensitive skin.
There are of course lots of other things that might be in your face cream, such as water, specific active ingredients, colours, fragrances, preservatives and UV-blocking sunscreens – but these are the three that make a moisturiser, a moisturiser!
References
J. N. Kraft and C. W. Lynde, Moisturizers: What They Are and a Practical Approach to Product Selection, Skin Therapy Letter 2005, 10, 1-8.
Z. D. Draelos, Therapeutic moisturizers, Dermatologic Clinics 2000, 18, 597-607.






love this post! very informative and interesting!
Thanks! 🙂
thanks! its so interesting how shape of such miniscule molecules give each chemical such different properties 🙂
Yeah, I love chemical structures 🙂
Cool, very interesting stuff. 😉
Thank you 🙂
I like this! And now makes more sense why certain ppl can’t use moisturises with whatever ingredients.
Thanks! This was really great
Thanks 😀
Yay science! This is very informative, thanks 🙂
Yay 🙂
Yay science! This is very informative, thanks 🙂
Hi Lab Muffin, love the informative science-bits. Could you say what, in your opinion, are the best (therapeutic) moisturising ingrediants to look for in a product i.e what REALLY works?
Many thanks and keep up the good work!
It’s hard to say, because it depends on what you want. From my experience:
– Mineral oil is often the only thing that will help you if you’re super dry and super sensitive. But for people who have oily skin to begin with, it can be too greasy.
– Shea butter is a really good emollient, amazing for rough patches. This works for me since my skin is usually very oily with a few dry bits – if a cream has shea butter as one of its top three ingredients, we’ll probably get along 🙂
For things apart from just plain moisturising (wrinkles, blackheads, sun damage etc), it gets a lot more complex…
Hope that helps!
Thumb up for your scientific explanations =)
I’m feeling smarter already -.- .
This has got to be one of my favourite posts on skincare, ever. You explained something I’ve been trying to understand for months SO succinctly and in a TOTAL noob-friendly way. Tbh the rest of your posts do the same. Thank you for sharing your wisdom T_T
I love the science. I wish product sellers knew their stuff like you. Many thanks!!! So I’m wondering about multipurpose products. Do they work etc. I’ve got combo with adult acne prone due to I believe hormones. Do you suggest three products for true best results or a wash that puts in water then perhaps two products for moisture. I’m on mission but do not know what to start with and yet so willing to start a new routine. Thanks for any help offered
Thanks so much for your wisdom sharing.
Thank you!
What are the effects of alcohol denat. within the top 5 ingredients of a product packaged as a “moisturizer”?
Is it generally a good thing or not to have denatured alcohol so high up on the ingredient list?
thanks
Alcohol is usually fine apart from dehydration – I should have a post on this soon! 😉