What is Olaplex?
Olaplex is a line of hair repair, “bond building” treatments that’s getting heaps of buzz in the hair community, especially with people who have damaged hair from excessive bleach.
It’s available in a couple of forms – Olaplex can be mixed in with bleaching products to minimise damage, or it can be used as a separate treatment. Here’s the science behind how it “repairs” disulfide bonds in hair.

Note (August 2018): I’ve updated this post, and there’s a video version now as well – click here to watch it!
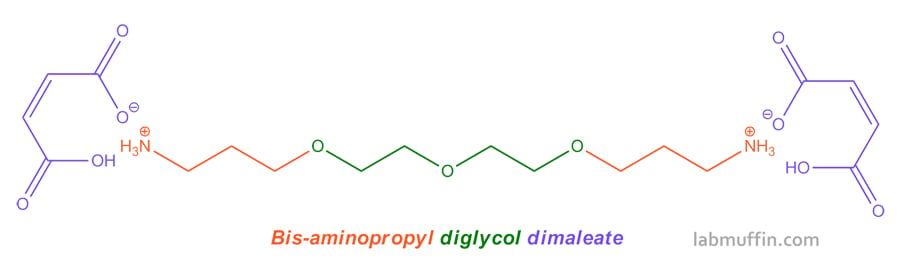
The active ingredient in Olaplex is a compound called bis-aminopropyl diglycol dimaleate. This is a slightly ambiguous name, but I’m pretty sure it matches this structure in the patent:
What does Olaplex claim to do?
Olaplex claims to “reconnect broken disulfide sulfur bonds in the hair.” The treatment is labelled a “bond multiplier”, which limits damage to hair during or after colouring.
Related Post: My Lazy Haircare Routine for Coloured Hair
A lot of people with damaged hair have managed to get amazing results from Olaplex. Here’s my friend Mary, who got her natural curl texture back with a single Olaplex treatment:
So suffice to say, it definitely does something! But is it as revolutionary as the hype makes it out to be?
How does Olaplex’s claims stand up?
Background
First up, a bit of basic hair chemistry. I’ve posted about hair chemistry before in my explanation of how hair straightening and perming work, but here’s a quick recap:
Related Post: How does hair straightening (and perming) work?
Hair contains lots of keratin proteins, which has the amino acid cysteine. Cysteine is special because it contains a sulfur (S) atom. Normally, two sulfurs will join together to form a disulfide bond (S-S), creating a link between two proteins:
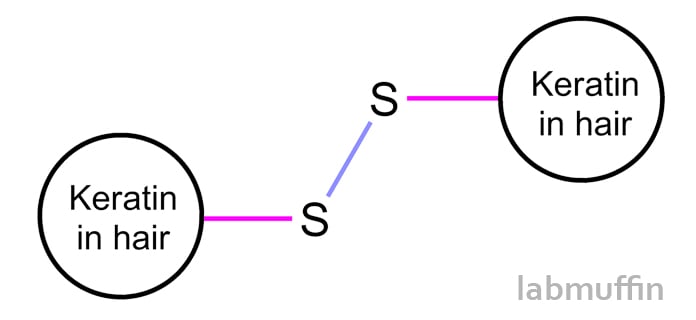
All these proteins holding hands is partially responsible for your hair’s overall shape and strength. When hair is permed or straightened, these bonds are deliberately broken into two SH (“free thiol”) groups, and then reformed after the hair is pulled into its new shape.
Re-forming these bonds typically takes a few days (hence not washing your hair for a few days after perming, since it warps the shape).
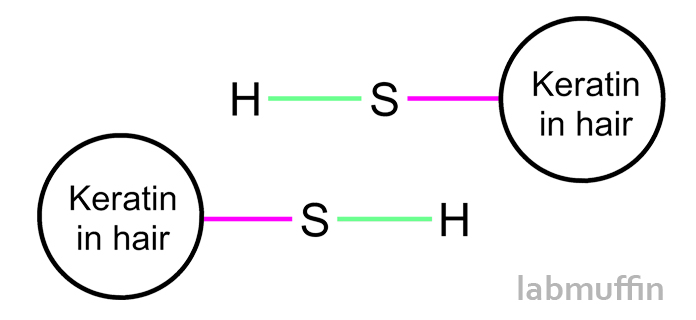
However, that’s not the only thing that can break disulfide bonds – high pH lye, repeated heat, exposure to sunlight, hair styling and chemical treatments like hair colouring will also break disulfide bonds and lead to weakened, damaged hair.
Related Post: Low Maintenance Pastel Hair Routine (video)
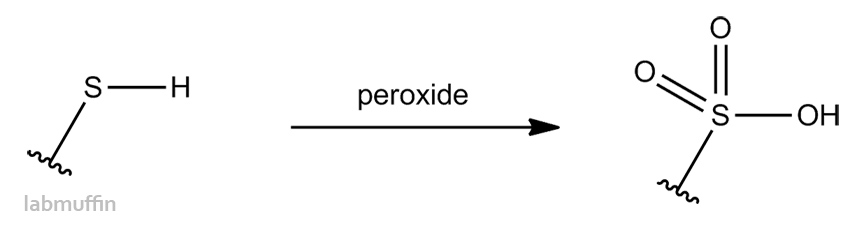
The peroxide used in bleaching can help repair disulfide bonds… but can also paradoxically stop them from forming, by capping the SH with a sulfate group, hence the extra damage:
Theoretical mechanism
I was initially sceptical of the claims, since so many other hair products claim to do similar things and…don’t.
But the patent holders are academic chemists with extremely impressive reputations – Craig Hawker is on the Editorial Board of a lot of high impact factor chemistry journals, and has 17 Angew. Chem. papers on his resume, which is a pretty BFD in the chemistry world.
So I wanted to vet this product thoroughly!
The patent has a surprising amount of detail into the mechanism of how it’s supposed to work. The short story is, the two ends of bis-aminopropyl diglycol dimaleate form bonds with the sulfurs, making an artificial, extended disulfide bridge.
Related Post: My Favourite Products from the Dermstore Sale
More specifically, the “dimaleate” part of bis-aminopropyl diglycol dimaleate are what’s known as Michael acceptors. These react with an SH group each in a Michael reaction to form covalent bonds like so:
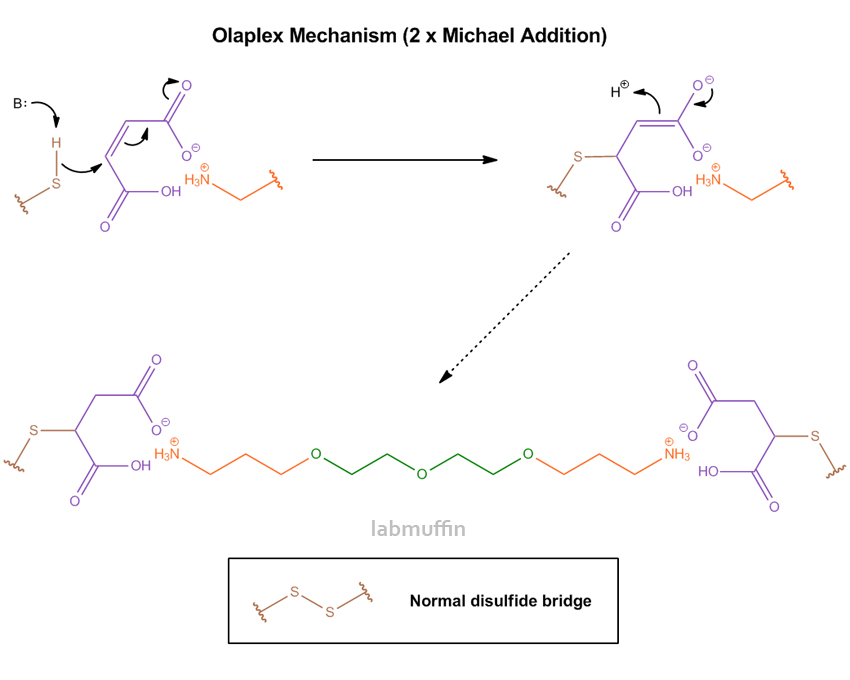 And so a new disulfide link is formed between the two sulfurs, making the hair stronger. This happens pretty quickly – unlike with hair straightening and perming, these reactions will happen faster than the peroxide capping reaction, meaning hair damage during colouring is drastically reduced.
And so a new disulfide link is formed between the two sulfurs, making the hair stronger. This happens pretty quickly – unlike with hair straightening and perming, these reactions will happen faster than the peroxide capping reaction, meaning hair damage during colouring is drastically reduced.
Pretty cool, huh?
In practice
It’s neat that there’s a scientifically legit explanation for how Olaplex could work, but does it really happen this way in the real world?
There are no published independent scientific trials yet, apart from thousands of rave reviews and before-and-after photos, which are pretty dramatic. There’s also a cool side-by-side comparison in the American Board of Certified Haircolorists Newsletter: they repeatedly shampooed hair after treatment with Olaplex, mineral oil, b3 Brazilian Bond Builder or colorpHlex.
Olaplex comes out on top by far, even though the other two proprietary treatments make similar claims (strengthens hair from the inside out, repairs bonds).
Olaplex is now routinely used in bleaching treatments, and can get black hair to white in a single session, without the hair becoming overly fragile!
I had my hair bleached from black to white (and then had purple applied over the top) at Ultrafade by Rich, a hair salon that’s well known for their amazing Asian hair transformations, and my hair is still in reasonable condition and can be heat styled without any breakage.

What are the different Olaplex products for?
There are 5 different Olaplex products now, and they’re imaginatively numbered 1 to 5:
Olaplex No. 1 Bond Multiplier
Olaplex No. 1 has the highest concentration of the active ingredient. It’s literally just water, bis-aminopropyl diglycol dimaleate and preservatives.
I’ve seen sources that say that it has 80% or 100% of the active ingredient, but if the ingredients list follows the usual rules, it can’t contain more than 50% (since the product has to contain more water).
It’s also very liquidy, which would not be the texture I’d expect for this chemical, although it’s really just a guess.
There are lots of ways that Olaplex recommend using No. 1: mixed in with bleach to reduce damage, mixed in with gloss, toner or colour, or diluted and used as the first step in a standalone treatment.
There’s a handy mixing chart on their site.
Ingredients: Water (Aqua), Bis-Aminopropyl Diglycol Dimaleate, Phenoxyethanol, Sodium Benzoate.
Olaplex No. 2 Bond Perfector
This is the second “professional use only” step that’s intended to repair any bonds missed by Olaplex No. 1.
Olaplex’s educational materials all highlight that Olaplex No. 2 (and 3 as well) are bond builders, not conditioning products – if left in, they won’t make your hair feel soft or nourished (in fact, they make your hair feel stiff and snaggy, as I know now from personal experience).
Instructions:
- Best applied to towel-dried hair
- Leave for at least 10 minutes (20+ minutes if possible)
- Shampoo and condition afterwards
- Shampoo before the Olaplex treatment as well if hair has build-up (oils, silicones, minerals)
Ingredients: Water (Aqua), Bis-Aminopropyl Diglycol Dimaleate, Propylene Glycol, Cetearyl Alcohol, Behentrimonium Methosulfate, Cetyl Alcohol, Phenoxyethanol, Glycerin, Hydroxyethyl Ethylcellulose, Stearamidopropyl Dimethylamine, Quaternium-91, Sodium Benzoate, Cetrimonium Methosulfate, Cetrimonium Chloride, Fragrance (Parfum), Polyquaternium-37, Tetrasodium EDTA, Butylphenyl Methylpropional, Etidronic Acid, Ascorbic Acid, Phytantriol, Prunus Amygdalus Dulcis (Sweet Almond) Oil, Tocopheryl Acetate, Aloe Barbadensis Leaf Juice, Panthenol, Simmondsia Chinensis (Jojoba) Seed Oil, Citric Acid, Potassium Sorbate.

Olaplex No. 3 Hair Perfector
Olaplex No. 3 is the take-home treatment. It has an identical ingredient list to Olaplex No. 2, but with a slightly lower concentration of the active ingredient (12.5% as opposed to 15% according to some sources).
I’m not convinced that a 1/6th decrease is a big enough difference to warrant it being labelled a completely different product with different uses – especially when both 2 and 3 are designed to be used on towel-dried hair, which typically contains enough water to dilute it by far more than 1/6th.
To me it just seems like an artificial attempt at differentiating “salon-only” “professional use” and consumer products, which is something the hair industry really enjoys doing.
Anyway, the usage directions are the same as Olaplex 2:
- Best applied to towel-dried hair
- Leave for at least 10 minutes (20+ minutes if possible)
- Shampoo and condition afterwards
- Shampoo before the Olaplex treatment as well if hair has build-up (oils, silicones, minerals)
- Use once a week, or 2-3 times a week for damaged hair
Ingredients: Water (Aqua), Bis-Aminopropyl Diglycol Dimaleate, Propylene Glycol, Cetearyl Alcohol, Behentrimonium Methosulfate, Cetyl Alcohol, Phenoxyethanol, Glycerin, Hydroxyethyl Ethylcellulose, Stearamidopropyl Dimethylamine, Quaternium-91, Sodium Benzoate, Cetrimonium Methosulfate, Cetrimonium Chloride, Fragrance (Parfum), Polyquaternium-37, Tetrasodium EDTA, Butylphenyl Methylpropional, Etidronic Acid, Ascorbic Acid, Phytantriol, Prunus Amygdalus Dulcis (Sweet Almond) Oil, Tocopheryl Acetate, Aloe Barbadensis Leaf Juice, Panthenol, Simmondsia Chinensis (Jojoba) Seed Oil, Citric Acid, Potassium Sorbate.
Olaplex No. 4 Bond Maintenance Shampoo
This shampoo has the longest ingredients list I’ve ever seen in a shampoo, and honestly I don’t know why any product needs this many ingredients.
Related Post: Does L’Oreal Colorista Hair Spray Work on Dark Hair?
It’s a sulfate-free shampoo, with bis-aminopropyl diglycol dimaleate as the 10th ingredient – mathematically this means it can’t be higher than 10%, but practically speaking, and considering the percentages in Olaplex 2 and 3, it’s probably far below that.
The amount of active ingredient seems a bit too low to be relying on this for repairing damaged hair, but that’s probably why they brand it “maintenance”. It does look like a pretty nice shampoo though.
Ingredients: Water (Aqua), Sodium Lauroyl Methyl lsethionate, Cocamidopropyl Hydroxysultaine, Potassium Cocoyl Glycinate, Disodium Cocoyl Glutamate, Sodium Lauroyl Sarcosinate, Potassium Cocoate, Decyl Glucoside, Glycereth-26, Bis-Aminopropyl Diglycol Dimaleate, Propanediol, Euterpe Oleracea Fruit Extract, Punica Granatum Extract, Rosmarinus Officinalis (Rosemary) Leaf Extract, Musa Sapientum (Banana) Fruit Extract, Origanum Vulgare Leaf Extract, Marinda Citrifolia Fruit Extract, Arctium Lappa Root Extract, Fragrance (Parfum), Cocamidopropylamine Oxide, Disodium Laureth Sulfosuccinate, Panthenol, Glycol Distearate, Sodium Cocoyl Glutamate, Methyl Gluceth-20, Acrylates Copolymer, Prunus Armeniaca (Apricot) Kernel Oil, Pseudozyma Epicola/Argania Spinosa Kernel Oil Ferment Filtrate, Pseudozyma Epicola/Camellia Sinensis Seed Oil Ferment Extract Filtrate, PEG-120 Methyl Glucose Dioleate, Amodimethicone, Sodium Lauryl Sulfoacetate, Guar Hydroxypropyltrimonium Chloride, Polyquaternium-10, Divinyldimethicone/Dimethicone Copolymer, Polyquaternium-11, Trisodium Ethylenediamine Disuccinate, C11-15 Pareth-7, Laureth-9, Glycerin, Trideceth-12, Hexyl Cinnamal, Limonene, C12-13 Pareth-23, C12-13 Pareth-3, Hydrolyzed Vegetable Protein PG-Propyl Silanetriol, Acetic Acid, Citral, Benzoic Acid, Tetrasodium EDTA, Pentasodium Triphosphate, Sodium Polyacrylate, Pentaerythrityl Tetra-Di-T-Butyl Hydroxyhydrocinnamate, Disodium EDTA, Sodium Benzoate, Potassium Sorbate, Phenoxyethanol, Chlorphenesin, Ethylhexylglycerin, Quaternium-95, PEG-8, PEG-8/SMDI Copolymer, Palmitoyl Myristyl Serinate, Citric Acid, Sodium Hyaluronate, Tocopherol, Biotin, Helianthus Annuus (Sunflower) Seed Extract, Helianthus Annuus (Sunflower) Seed Oil.
Olaplex No. 5 Bond Maintenance Conditioner
This is a low-silicone conditioner with a very long ingredients list again – this time bis-aminopropyl diglycol dimaleate is ingredient number 14 (if you’re still following along mathematically, it’s less than 7%).
Again, the amount of active ingredient looks a bit low for using it “as Olaplex”, although it does look like a nice conditioner.
Ingredients: Water (Aqua), Cetearyl Alcohol, PPG-3 Benzyl Ether Myristate, Caprylic/Capric Triglyceride, Cetyl Alcohol, Octyldodecyl Ricinoleate, Quaternium-91, Cetrimonium Chloride, Divinyldimethicone/Dimethicone Copolymer, Behentrimonium Chloride, Glycerin, Cetyl Esters, lsododecane, Bis-Aminopropyl Diglycol Dimaleate, Fragrance (Parfum), Panthenol, Phospholipids, Dimethicone PEG-7 lsostearate, Pseudozyma Epicola/Argania Spinosa Kernel Oil Ferment Filtrate, Pseudozyma Epicola/Camellia Sinensis Seed Oil Ferment Extract Filtrate, Tocopheryl Linoleate/Oleate, Quaternium-95, Propanediol, Punica Granatum Extract, Marinda Citrifolia Fruit Extract, PEG-8, Euterpe Oleracea Fruit Extract, Camellia Sinensis Seed Oil, Crambe Abyssinica Seed Oil, Hydroxypropyl Cyclodextrin, Persea Gratissima (Avocado) Oil, Vitis Vinifera (Grape) Seed Oil, Disodium EDTA, Polysilicone-15, C11-15 Pareth-7, Hydroxypropyl Guar, Glycine Soja (Soybean) Oil, PEG-45M, PEG-7 Amodimethicone, Amodimethicone, C12-13 Pareth-23, C12-13 Pareth-3, Laureth-9, Pentaerythrityl Tetra-Di-T-Butyl Hydroxyhydrocinnamate, PEG-4, Phenoxyethanol, Hexyl Cinnamal, Limonene, Trideceth-12, Sodium Polyaspartate, Citral, Potassium Sorbate, lodopropynyl Butylcarbamate, Silica, Acetic Acid, Etidronic Acid, Sodium Acetate, Chlorphenesin, Propylene Glycol, PEG-4 Dilaurate, PEG-4 Laurate, BHT, Sodium Dehydroacetate, PEG-8/SMDI Copolymer, Palmitoyl Myristyl Serinate, Sodium Polyacrylate, Ahnfeltiopsis Concinna Extract, Sodium Benzoate, Citric Acid, Tocopherol, Hydrolyzed Hyaluronic Acid, Biotin, Helianthus Annuus (Sunflower) Seed Extract, Helianthus Annuus (Sunflower) Seed Oil.
Further Questions
I had a few questions about Olaplex based on the mechanism of how it works – these are very speculative!
Could Olaplex be used for perming?
Since this method forms disulfide bonds much faster than traditional methods, could Olaplex be used for perms where you can wash your hair straight away?
I personally can’t see why not, though perhaps it hasn’t been marketed that way because it’s less revolutionary.
Why are there ionic bonds in the structure?
The maleate sections of the molecule are joined to the linker by ionic bonds (the attraction between the + and – bits), rather than a covalent bond (a solid line).
Covalent bonds are generally stronger than ionic bonds, and generally ionic bonds are more susceptible to being broken if there’s a lot of water and other ions around or if there are pH changes, which you’d expect with regular shampooing.
Why did the inventors choose to use ionic bonds?
Is it because the ionic bonds used here are unusually strong or have slightly more angular freedom (covalent bonds are like rigid welded joints, while ionic bonds are a bit more like ball-and-socket joints)?
Or is it so the effects of Olaplex will wear off faster and you’ll have to use more product?
Is it planned obsolescence, so they can unveil Olaplex Permanent 2.0 in a few years time, with covalent bonds where the ionic bonds are?
(It’s worth noting that the patent covers versions with covalent bonds, and currently the effects are estimated to “dissipate in a few weeks to a month” – the patent mentions “two months or more”.) I don’t think I’m going to get a good answer to this anytime soon.
Is there any danger of the hair becoming harder to repair, if it does get damaged after Olaplex?
This question is mostly idle speculation on my part, and it could well be a silly question.
With normal hair, the weakest list is usually the disulfide bond. When it breaks, is becomes free thiols, which can be repaired with oxygen or peroxide (very slowly and unreliably) or Olaplex (quickly and probably reliably).
But what happens when Olaplex comes into the picture? I suspect the ionic bond will be the weakest point, and you’ll be left with this on the ends:
Will this act as a “cap” to stop further disulfide bridges forming, preventing further Olaplex treatments from working?
TL;DR version
From a chemical perspective, Olaplex could definitely work to repair hair in a way which no other product on the market currently does.
Unfortunately I can’t find a list of salons in Australia with Olaplex, but it’s distributed by Haircare Australia, who may be able to help.
There are also some sellers of Olaplex on Amazon.
Here’s the video version – click here to watch it on YouTube.
Have you tried Olaplex? Does it live up to the hype?
This post contains affiliate links – if you decide to click through and support Lab Muffin financially, thank you! For more information, see Disclosure Policy.








Isabel/Ruben,
I run the international division of Olaplex.
If you need any more technical information from Greg or Eric they are happy to answer questions you may have?
Let me know & thanks for a fine piece of objective journalism.
Kind Regards
Ed Connaghan
Thank you Ed! I’ll get in touch 🙂
How would you use Olaplex as a perm? Could it be used to permanently straighten hair? Thanks!
How would you use Olaplex as a perm? Could it be used to permanently straighten hair? Thanks!
You use it in the perm of choice not as the perm.
You use it in with perm of choice not as a perm
You would not perm hair with it.. But added to perm solutions would help to repair bonds that would be compromised by chemical damage x
can i have some infor on this product and are you looking for a trail salon in Queensland Australia
Do you own a salon in Australia?
We’ve been Trialing all the ‘Plex’ versions in our Salon: http://www.lilyjackson.com.au.
In our trials we have used (Olaplex, Cureplex, Snapplex) these treatments, in our experience with them only benefited hair that is already very damaged.
If the hair was virgin/healthy- our experience showed us that- We wouldn’t recommend these products -They slowed the colour development and affected the end result, in some cases the colour development didn’t work at all!
If the hair is fine and healthy the cuticle layer is very tightly shut and a ‘Plex’ treatment will add a barrier and we have seen a bleach application with 12% barely lighten the hair.
In my experience during this trail with these products they have a limited place but it is not a Miracle cure that Marketing would have it be.
The Plex’s ingredient if over used can make the hair feel brittle, was something we defiantly saw in our Trial clients hair
It does seem penetrate the cuticle layer and the ingredient seemed to act like a support system for the disulphide bond, this however is not permanent and it
left the hair and the damage already existing remained in all the cases we saw.
But Like I said if you have very damaged hair and you want to push it some more, the products may add a level of protection in these cases.
We recommend to our clients to use these products with your eye’s open and know the facts before committing to the treatment with your stylist. ie Do you Really need it?
In Sydney these treatments start at $50-$80 on top of your colour service, mostly because the Australian Distributors are cashing in on the new Buzz service and have tripled the wholesale cost of particularly of Olaplex.
My opinion- for what its worth- investment in a great colourist / stylist that uses high end Colour technology (not low grade ingredient brands) as these products will work against damaging your hair as they colour it.
I have had the in salon treatment and the take home bottle. I know there are loads of people raving about how great this is and it’s being hyped as the next big thing in hair.
My experience was just negative. I was sceptical so if there had been no perceptible change I would not have been compelled to comment. Just put it down as another miracle product that didn’t work.
However, My hair actually felt worse. There was a change in texture but not in a good way, more like there was product left in my hair.
I also noticed when my hairdresser dried it, it wasn’t as straight as in would normally be in salon. When I dried it it was even worse obviously!
It was also just as prone to breakage the whole time, so there was absolutely no difference there. I would honestly suggest you err on the side of caution. This cost me over £55 and I would definitely not use again. I wasn’t trying to get my hair lighter, my hairdresser recommended it because of breakage to strengthen my hair between colours.
One other thing I also noticed was my hands would be very dry on the day I used this. There was enough to use once a week until my next appointment but a only used it 4 times and then refused it at the salon when I had my next colour. it would be marvellous if it did work, maybe it does for some but definitely not me!!!!
The same happened to my hair and I feel for you!! The company doesn’t want to take Responsibility for the damage the product is causing…they just want to say to everyone who is complaining that the stylists do not know what they are doing!! Please do not blame you Salon or the Stylist!! You are not alone, I have found people all over the internet that has the same issues!! You will see The Truth will come out!!
I noticed the same….. Do you know how long the treatment lasts in hair?
I have no idea, I had to cut my hair off…I wish I had an answer for you and i feel for you! There just isn’t any answer and the company is just sticking their head in the sand and blaming the stylist or the customers not following directions…etc…Their wrong!!
My experience was the same
I’d heard all the hype and waited for it to come to Australia and when I finally tried it my hair felt dryer and very porous/brittle etc
when I told my hairdresser she told me to up the treatments to twice a week and it got worse with a fair amount of breakage I decided to stop using it and went back to the coconut oil treatments and my hair is finally feeling okay again but I’ll never use olaplex again
I have the same thing…. Change in texture and not in a
good way. I used to have great body in my hair. Now it is actually finer and has no body whatsoever especially on the day I don’t wash it. If I use extra product for body it feels really dirty and like there is too much product. .. And,….. It is EXCEEDINGLY more brittle. I wouldn’t recommend this to any one.
I wish I could find someone who has had it done several months ago to see how long this effect will last in my hair. …… 6 weeks?…6 months?…… I feel like just wearing it in a clip everyday now. So upset. I have asked hairdressers and even they don’t know the answer…………!!!
I’ve just had the salon treatment (stages 1 & 2), my hair felt really good on the day it was done, but after the first home wash, I can’t really say that it feels any different to how it did before. (I have quite thick, naturally curly/frizzy hair)
Thinking of buying the stage 3 just to see if it helps, but I don’t think it’s a miracle treatment, which many people seem to claim.
Thank you for comment, I was just told about this.
My hair also 6 days later washing 3x in the 6 days always feels like product is on it when there is none i m getting so mych breakage while combing it wet .
Olaplex can have negative results of you have any build up on your hair from water products etc. As for changing the texture the 2 yrs it’s been out this first time I’ve heard that one. Do you have a natural wave or curl pattern in your hair?
Maybe the application was done improperly.
I felt exactly the same,only paid €20 though
I can not speak on the other brands but I have used Olaplex. I have had hair feel better after lightening than it did before we started and this is using 30 and 40 volume for up to an hour. And as far as barely lightening with 12 volume I would expect that. If you follow the directions and dont exceed what the hair can handle you too could have great results. For adding to color I dont exceed an 8th of an ounce per ounce of color. And if you are using with lightening you need to increase your volume by 10 for the result you are looking for. If you are looking for a 10 volume result you need to bump it up to 20 volume and so forth. So if you used 12 volume that would be in anticipation of a 2 volume result so if it barely lifted that would be correct.
I believe the lady above was referring to using Olaplex with 12% hydrogen peroxide, not 12 volume. 12% is 40 volume.
I think you are so right I had opalex twice & felt my hair was definitely drier that was it…total waste of money. Salons are pushing it lijevits the holy grail & it certainly is not….
It doesn’t slow down colour development. In my experience as a Wella MCE, unpredictable, bad, worse, unusal colour results or development always stem from the user, not the product. This is true of ALL big brands of colour, or at least all the tried and tested ranges – Koleston, Majirel, Igora Royal etc.
As for people saying the hair felt worse or that there was a residue, again you blame the stylist not the product.
More importantly, though, you and the stylist must understand what Olaplex does. MOST important, more than knowing what it does, is knowing what it DOESN’T do –
IT IS NOT A CONDITIONER. IT DOESN’T SOFTEN THE HAIR OR ADD MOISTURE OR PROTEIN.
Why is this so important? Yes, you need the bonds to be connected, but chemicals and environmental factors also strip the hair of its protein and moisture.
OLAPLEX DOESN’T REPLACE OR TAKE AWAY THE NEED FOR PENETRATING CONDITIONERS.
This is very important to understand because you can use Olaplex to prevent breakage and to improve hair integrity and also help colour to last longer – I use it constantly with great results.
The problem is the word “condition”. There are different ways something can have good or bad condition. as an analogy, imagine you stitch together a load of scraps of dry leather to make a jacket. Yes, it is together and the stitches are strong, so it is in a good state in that it won’t fall apart. It is in “good condition”. BUT, the leather is still dry and stiff. One would need, also, to treat the leather to have it supple.
This analogy, with the stitching relating to Olaplex and treatment of leather for suppleness relating to condition treatments (penetrating conditioners) represents the hair.
I am wanting to get my bleached hair chemically straightened – I have tried Keratin but it does not work on the strong waves I have under the top layer of my hair & I was told after contacting Olaplex Australia that it would work on my hair. I was still not sold as I’ve heard so many hair salons use it as yet another money making scheme & after reading the above comments how can anyone find a hairdresser they can actually trust to do the job properly?? Ofcourse all the salons that use it are going to give positive feedback to Olaplex, if it is making them money?? I desperately want to get my bleached hair chemically straightened but after reading so many bad reviews how can I?? At the risk of having hair left like chewing gum or simply breaking off – my question is if Olaplex is blaming the user & there are so many bad reviews, how can anyone ever decide where to get their hair coloured, straightened or permed??
You hit the nail on the head with your comment. Absolutely right.
You hit the nail on the head
I live in Sydney do you know where I can find the product?
Very informative response, thanks! Good to know..
You are very smart!! Try Mondo Verde Hair Care. I’m in Houston at All Access Beauty Supply starting January 1st
The alternative linker you propose is propably water unstable
Hi Michelle!
I didn’t know where to leave this comment but this’ll do. I read your article which was very insightful. But, typically, most ionic bonds are actually stronger than covalents. You’d need to actually measure the bond energies of each, along with electronegative properties to decide but it’s usually the ionic the strongest. They form a tighter lattice structure. The ionic definitely have higher boiling points and melting points and they move less compared to covalent bonds especially with single bonds that are always creating dipole to dipole moments which break easier.
Thanks for your comment!
For these bonds though, the cation is an ammonium – I think they’re generally weak? (since ammonium compounds are pretty soluble in water).
I don’t know what you are on about, but I paid £400 for 2 sessions of lightening my hair with olaplex. The first was 6 hours long and three applications and the second took 3 hours. My hair went from v dark brown to ginger. Then the texture changed to ‘soft fuzzy curly” and then it started to break and 3 months later it has broken and thinned by half!!! I had very long hair and its now below shoulder but thin and broken and horrible. Vi wish I had NEVER used olaplex. It has totally ruined my hair. It’s shocking and I don’t know who to turn to.
Olaplex did not ruin your hair. It might be the reason you have any hair left. Olaplex should be used to mitigate any damage that will occur from lightening your hair. I dont know why any stylist would have subjected you to lightening for 6 hours and 3 applications all in one session. The very idea makes me cringe.
I have had a similar experience, however I don’t think the Olaplex is at fault. I wanted to lighten my dark hair to a medium ash brown, and had to lighten it over 3 sessions to try to get the ash colour I wanted. I had hoped that the Olaplex would prevent any major damage. I waited 2 weeks in between each lightening and used/still use the “at home” Olaplex option.** I stopped using Kerastase products and treatments at this time to investigate Olaplex. Its been about 8 months now since i did this and I find my hair getting more dry and brittle over time. I would say the damage is from lightening, not the treatment itself. I increased the frequency of treatments but find they only last 1-2 washes and then my hair is a tangled dry dull mess again. In my opinion the Olaplex may have helped “control” the immediate damage but certainly did not prevent it, and in the long term, the damage is going to become evident. Today, out of frustration and memories of how my hair used to feel before switching to Olaplex, I returned to a salon to get a double boost of Kerastase moisture and strengthening treatments and ..WOW I have my old hair back! 🙂 I know it hasn’t repaired the damage, but at least it is smooth and glossy and so much more full of life! I typically could go about 8 washes before needing another Kerastase treatment so it will be interesting to see how long this lasts, but i can honestly say today I am the happiest I have been with my hair in a very long time! I will be using both Olaplex and Kerastase in the future 🙂
To me it olaplex isn’t at fault. I use olaplex every day of my working life. It seems some hair stylists are forgetting all the rules of hair colouring when using olaplex as an additive. Colouring hair 3x in a day with or without olaplex will have a drying effect on the hair. Lifting slowly & in sessions is always the best way. Thinking olaplex is a magic wand & hitting the hair hard with multiple lightening sessions then blaming the additive!
I am a 26 yr veteran in the industry. I’ve used Olaplex for the last 6 times I’ve colored. I use Joico 5XR and 20vol. I only color my hair once every 3 months or so. My hair is naturally curly/wavy, super fine, and I don’t heat style except my bangs.. Blow dry only. I use Pureology hydrate.
Olaplex left my hair so brittle and dried out that no conditioner or treatment had any effect. I had to cut nearly 5″ of destroyed hair that was unfixable.
I have seen it keep hair together, I’ve seen hair pushed to outer limits. It might still be attached, but in my case it was horrible. It doesn’t work for everyone. It’s a bond multiplier, not a miracle end to damage.
As with any product you should strand test before putting all over your hair. It may not be for everyone or certain types of condition of hair.
Olaplex has never claimed to be a miracle lifter! It’s a tool to lift and help the hair. YOUR STYLIST is the one to be upset with. The stylist are the one to use discretion when doing big jobs such as lifting dark to blonde. Usually recommend to do over numerous sessions. It also has a lot to do with home care as well. Products heat that’s applied etc. Just saying Olaplex is worth every dime to my clients.
I’m curious, did you ever get answers to your questions about the choice of ionic bonds in the structure and if the hair becomes more difficult to repair with continued use of Olaplex? Have you used it for yourself and formed any opinions about it? I had a olaplex treatment in the salon (separate from any other service) and bought a bottle of the take home treatment, and I’m a fan. I dye my hair a darker color, and after doing so my hair feels rougher in texture. I applied the take home treatment to my hair afterwards and my hair was much shinier, softer, and less frizzy than even before I dyed it! I’m just curious about your thoughts about it now that it has been out awhile. I’m curious about potentially relaxing my hair (chemically straightening) but I would never do it without Olaplex.
I appreciate the technical information you included vs the typical “it repairs the bonds” type review. I wrote a paper, although not nearly as technical and with much less education, many years ago explaining how a “perm” breaks down hair bonds and the bonds reform to create the new shape of the hair.
I understand formula 1 is used during the bleach or coloring process but sev stylists have started Olaplex treatments in their salons for damaged hair, applying Olaplex 1, followed by adding Olaplex 2 after approx 30 minutes.
My question is related to the ability of Olaplex 1 to penetrate the hair when used without color or bleach. In your opinion, would using a low volume peroxide, such as volume 5, help open the hair for the product to fully work? Or would the additional developer cause more damage to very badly damaged hair?
I realize the post I’m referencing isn’t recent. I hope to find additional info from you!
Thank you so much!
I honestly don’t know, it could go either way! I think you’ll get a good answer from an Olaplex rep or a hairdresser who’s tried both.
I’m replying because your post is recent. My stylist has used Olaplex twice on my highlighted hair. This is the only change I’ve made…. no other new products or styling tools. No medication. My hair is wrecked. I have just below shoulder length hair and I now have large sections that have broken off to approximately 2 inches from my scalp. My hair is so dry and thin I’m afraid to brush it. It is NOT my stylist. She too, is unchanged and over the 35 years she’s been doing my hair, it’s never been like this. I’m considering litigation so I may bring attention to this farce in order to keep others from experiencing this horror.
The exact same thing has happened to me after using olaplex ?
Olaplex has literally been my worst nightmare for my hair. It turned my soft, shiny, smooth hair into rough tangled crinkly strands with a SINGLE STANDALONE TREATMENT. NO BLEACH OR COLOR. I left it on for 4 hours and I remember the panic as I rinsed it out my hair felt like rough (like holy crap ROUGH) and so knotted together and it completely stripped all shine and it feels like its coated in something and had lost all volume. Its been over 6 months and I still want to cry because my damaged hair is breaking more and more.
The product should seriously have more warnings on it. This Jordan guy is totally delusional or ignorant to think it can’t damage hair when so many people have complained about the same disaster I have experienced. I struggle to style, brush, manipulate my hair in any way. I want my pre-olaplex hair back sooo bad. Patience girls, just gotta let it grow ?:)
I have been using bond maintenance conditioner N.5 and bond maintenance shampoo N.3.i recommenced this from my hairdresser who I have been going for 25 years but it hasn’t quiet worked out for me.it has been very frizzy and I have had complications during this process of healing the brittle hair and frizzy can you help me
hello. I have a question.. my hair is very damaged and was so dry and breaking it would stand up on its own.. YES THAT BAD!! We did an Olaplex treatment on it and it feels AMAZING!! How often can I do a treatment on my hair.. and will back to back treatments even help it more?
thank you,
Angel
Yes, you can do back to back treatments. I am a cosmetologist currently we or king with Olaplex snd am loving the results I have been getting. Make sure you shampoo and deep condition after the treatments. You need moisturd, pfot ed ins, and keratin added into hair also to help maintain and add health back into the hsir. Olaplex is definitely amazing!!!
Hi I have dark very damaged hair. I would like to stay dark but also help my hair. Can I use opalex? Or will it turn blonde? As only ready about blonde hair use. Thanks Caroline
Olaplex can be used on all colors of hair
You may do treatment as often as you’d like. Olaplex is not a protein and cannot be overused.
I’d do it once a week but still use a high quality conditioning treatment
What about using olaplex with well water? I’m curious if there has been any research or a theory of how it should and shouldn’t react. There are several residents here in North Carolina that are using well water.
The only effect that happens when there are minerals in the hair is that Olaplex is unable to penetrate through the buildup. Any sort of mineral “reaction” is caused by lighteners/color and the minerals themselves. This is something that has been happening over the years and is not caused by Olaplex. I also want to stress that if you do see this sort of reaction, to rinse immediately because although Olaplex is an insurance for the hair, it will NOT prevent damage from this type of chemical reaction. Olaplex is formulated to mitigate the damage from the breaking of bonds caused by lightener/color/ perms etc. If you have anymore questions, please Email me at Ab@Olaplex.com
can you further clarify this? How would one know if there were too many mineral deposits in the hair? If it’s treated for that first, and then with olaplex, would that be safe? This comment sort of scared me that olaplex could react badly if there is mineral buildup, and cause irrepreble damage….?
I clarify everytime before use as there’s no certain way to know
I have well water and have had no problems for the last five years. I recently had application of olaplex and my hair felt dry and I experienced some breakage, more than normal with my color treatment. My hairdresser contacted oloplex and they said it would not cause these problems. I just went for my second treatment and ended up with major breakage, dry brittle hair and all kinds of fuzzy breakage. I went back to hairdresser 1 week later to get a conditioning treatment. He could not believe how damaged my hair was in one week. I have long hair and the fly away fuzz & breakage starts at the ends and goes all the way up to my root. Your company claims that it cannot be the olaplex, but nothing has changed. My hair does not feel or look like my hair did prior to these applications. We contacted the company that treats our well water regularly to have them test water, they found no problems with the ph or softeners. The only difference is olaplex. Have you seen this in any other clients? And, how would you know if I have mineral in my hair?
Hello, I just wanted to say that I have also had a bad reaction to Olaplex. Here is the main thing. This product is not a permanent bond, in fact nothing is. This product is a temporary bond and will result in breakage either at the bowl or later at home when the bond is washed away. I’ve been a bleach and tone for 10 years. NEVER have I had breakage like what I have now AND adding this Olaplex is the ONLY thing that was different. It increases the process time and in time with other clients salons are seeing this damage and people will realize this product is no miracle. I regret falling for the hype on this. Time is key here. Other people are saying this same thing. I would rather have my service done in a reasonable time frame and adhere to good home care rather than have someone push this product only to break later. NOT worth it.
“Olaplex” did the same damage to my hair! They told me the same story they told you, that it can not create any damage!! I only used hair color Level-8N 1/2 with Level-9G for 25min. pulled through 3-5min. at max…My hair texture is fine just like yours but, I had a lot of it…All gone Now!! They do nothing but bully you and said I didn’t rinse all the color out…Wrong!! They are delusional and we will see in the future “The Truth Always Prevails”
Call my attorney Janet Varnell @ 352-753-8600 She’s handling the “WEN” class action suit!!
I’ve had the same bad experience with Olaplex. Breakage, hair loss and just overall it seems to have lost it’s body. I’ve been getting highlights in my long hair for years and always took good care of it. Last winter my hair stylist advised me to try Olaplex. At first my hair looked fine, not better, not worse. I took home the step three and started using it according to the instructions. But after two weeks I noticed that my hair started to become really dry. It became so brittle that it started to break off. I’ve noticed that my hair is thinner and the structure of it seems different than before. I’ve never had this with other color treatments. Now, a few months down the line, I’ve had to chop most of my hair off.
I honestly wish I had never used this, the only thing different in the equation was the Olaplex. I have no idea how long it will take for my hair to get back to it’s natural condition. If people are reading this and have found a way to get their hair in good condition again after using Olaplex I would love to hear it.
Anyway, if you want my 2 cents, the product does not deliver and there are too many people who are having the same problems. So I wouldn’t take the chance of ruining your hair.
All well water has certain minerals in it that cause a chain reaction. That’s why it’s recommended to clarify before treatments
Do your shampoos, shower gels and soaps lather easily? If not, you likely have hard water which has mineral deposits – this is the nature of hard water.
I was told by friends with well water that their water behaves as hard water and has lots of deposits – a bit like bottled mineral waters come from underground sources.
I wonder if all the other negative reviewers have hard water, too?
I live in a soft water area and Olaplex has made a big difference to my hair. I colour my hair at home and only use the No 3, but follow the instructions and condition after use.
Hello Ed,
I have a medical question regarding Olaplex for Eric or Greg.
Would email be an option to get in touch?
Thanks, Debbie
OLÁ ED. RESIDO NO BRASIL ONDE NÃO HA REPRESENTANTES DO PRODUTO OLAPLEX,TENHO GRANDE INTERESSE EM ADQUIRIR, GOSTARIA QUE ALGUM REPRESENTANTE ENTRASSE EM CONTATO COMIGO PARA QUE EU POSSA REALIZAR A COMPRA. DESDE JA AGRADEÇO
Ola Gilmara,
Gostaria de saber se algum representante ja entrou em contato com voce, ou se voce ainda precisa de ajuda.
Obrigada
How do you use olaplex with a perm?
Is this s product good for only color treated hair also is it recommend for African American hair. If so what is the proper way to use it with a relaxer & just as a conditioner.
Can you get Olaplex put in your hair to repair damaged hair (split ends, dry hair) or is it only to repair bleach damamged hair?
Thank you
Olaplex will repair bonds in hair structure. Can be used in any type hair. Just make sure you clarify the hair first to remove any build up and minerals. I comb step 2 for about 10 mins through the hair than let sit the last 10 I’ve had nothing but 5 star ratings from Olaplex in almost 3 yrs I think all the complaints are the product not being used properly!!!
I like to know more about this product. Can you use this after color
You can use before during and after! Always clarify hair before a treatment to remove any unwanted build up. It helps the color last longer the colors are more vibrant and if done properly hair is like silk!
How do you ensure it is done properly?
Hi I am a hairdresser 34 yrs some one tried to sell this product to me to day I would like more info if you can provide so details about the products
Steve chandler
How often can I use olaplex ? I used it last month . Is it too soon to do it again??
You can use it as much as you like. Daily weekly monthly just make sure clarify hair before to remove any kind of build up
I have very dry damaged wavy hair. I am allergic to MMA methyl methacrylate, acrylates and copolymers. It this treatment safe for me?
Hi i would like to buy the product , can send me some information
Thanks
Did you go to Olaplex.com? If you want product information you should go to that product’s website 1st for that information. This article is headed by Lab Muffin not Olaplex. She didn’t state that she works for Olaplex. Therefore, if you want information to read or want to ask a question about the product it’s best that you go to the source.
Have there been peer reviewed scientific studies of this product that prove it, literally, repairs damaged hair, as if the damage never took place? How long does the “bond repair” last? How does the product “know” what exactly it needs to “repair”? What does it do to the bonds that aren’t broken? If this product does what it claims, what are the other possible applications, such as it’s use on fingernails and toenails? What kind of damage can this product produce in hair, side effects? All in all, I suspect there are no peer reviewed studies of any of your products which, therefore, suggests your company may have a bit to hide, Mr. Conohaghn.
The actual article answers half your questions. You should read it first.
I have to ask WHO ARE YOU ASKING? Cause the creators name isn’t what you put
Seem a lil upset that it’s a brilliant invention that is patented and millions of unpaid stylists and other stylists support it
These are still valid questions. If independent studies prove the value of Olaplex then even more people will want to try it.
Hi my email is rachelestelle75@ aol.com can you forward all scientific information ? Many thanks Rachel Bell
I was reading through the comments and felt i should share my experience with Olaplex.
I had stage 1 and 2 done at the salon a week ago and bought stage 3 for 1x week at home.
BEFORE having the treatment done, my hair was severely damaged from over bleaching and very gummy when wet, i could not even touch my hair til it was dry. I am an older woman (50) and i am sure this also has some effect on hair texture.
AFTER the treatment my hair feels thicker, heavier ( like when i was a kid ) and very soft. i can run my fingers through it while it is wet ). I absolutely love the results and i hope that my weekly home treatments and every 2 month salon treatments will make my hair continue to feel like this. I want to grow my hair long again ). oh, and i was my hair daily. This is my experience so far.
Ed can you use olaplex of you are pregnant ???
Hi , can you just use no 2 olaplex by its self as i do not need to use number 1 , Thank you Mandy .
May we find out about the company behind Olaplex? Was it set up solely for Olaplex? Or did you produce and market other products before Olaplex? How long has the company been in existence?
Thank you very much
Hi i have very damaged bleached hair that is breaking and extremly dry i have done two treatments of olaplex now and its still really damaged ive just purchased 100ml of each and was wondering roughly how many treatments i need to do before my hair is repaired? Also can i do 1 treatment a day to speed things up?
Thank you roxanne
Can you use opalex no 3 without 1or 2 and will it still repair damaged hair thsnkyou
Yes. That’s what I do. I use it about once a week according to the instructions (although I try to leave it on as long as possible, sometimes several hours) and I get great results. My hair is very soft and shiny afterwards.
I am a chemist, too, and I would love to know about the reasoning for ionic vs. covalent bonds as well! Will the covalently-bound product be released, too, or was that just a necessary precaution in the patent? Is there something about using the ionic bonds with maleic acid that makes it a better treatment? thanks!! 🙂
Another chemistry question: is there anything in the step 2/3 formulation that is chemically required to make the step 1 crosslinking work optimally?
I love this blog by a PhD chemist! makes me so happy 🙂 I am working on my PhD in chemistry too!
Yay!!! My 14 yr old daughter has been pressuring me to use green hair dye. I feel more comfy regarding uses an option.
Ed,
as head of the international division of Olaplex. I have a simple question that I am hoping that you may be able answer. Some reports state the No 2 treatment should stay on for 20 minutes, others that it should stay on for 10 minuets. I am not a professional hairdresser, not even an amateur hairdresser but I need to know from start to finish how much time should it take for a competent qualified hairdresser to complete the basic Olaplex treatment. That is using the No 1 and No 2 treatments. As a guide would it be 30 minutes, 45 minutes, one hour, one hour fifteen minutes or even one hour 30 minutes. or even longer than that.
Perhaps you would be kind enough to ask one of your staff to provide this information as it is important to me.
I look forward to receiving a favourable (favorable) reply.
Brian Massingham
e-mail…….brianmass2@yahoo.co.uk
Can i use olaplex step 1 and 2 at home as a treatment and then go to a salon to do my highlights because i salon doesn’t use this olaplex. I want to regularly do i at home as a treatment and then do the procedure at the salon like this will olaplex still work and condition my hair. Ty
Connie
is it different from keratin treatment?
I have only just come across this product , all reviews seem amazing
I use 30 or 40 volume developer an bleach powder to lift my hair, it never lifts as light as I would like. Will this product help? Thank you
actually any oil made to use with bleach when added will lift much more.
I recently tried Tressa Watercolors bleach lightener. I use it at home because they say no more than 20 volume and no heat! And it really does work. However I am not using it at salon because I process my colors with steam and am afraid that the Tressa will go too light.
Does this mean that someone with relaxed hair (via chemical relaxer) can get a perm if Olaplex is used during the perm process?
Can I use olapex on rebonding damaged hair?
Hi. I just had a treatment at a new hairdressers. He insisted only bleach would lighten my hair. I’m pretty sure my other hairdresser uses high lift and never bleach. I went with him because he uses olaplex.
It was my understanding the olaplex is applied during treatment, but he applied it after the colour/ bleach was rinsed out. He did say if he applied it during treatment, he would need to use a stronger bleach and I would walk out with my hair in a bag!
My hair is naturally dark blond/ light brown, thick and in good condition, just a little dry. I’m getting married in two months and wanted to try the olaplex to help my hair. I was nervous about the bleach, but he insisted my other hair dressers would all have used bleach.
I’ve washed my hair two days later and could cry. It is sooo frizzy and fluffy and dry. I am wishing I had not bothered with olaplex and stuck to my usual hairdresser!
I am no professional but even I, after five minutes of research prior to using olaplex at home with my lightener/bleach I know that Olaplex No 1 is added into the lightender/bleach (3,75 ml for 15 g of powder lightener/bleach) where you need to up the developer by one level (9%/30 vol instead of 6%/20 vol). Then after rinsing out the lightener/bleach, Olapex No 2 is put on the hair for 10 min (I left it on for 20 minutes myself). Then the Olaplex No 2 is washed out and the hair shampooed and conditioned.
Your hairdresser apparently hasn’t read the instructions. Olaplex No 1 weakens the developer by one level. So him saying that adding Olaplex No 1 to the bleach would harm the hair since you have to up the volume is nonsense since with Olaplex a 9%/30 vol works like a 6%/20 vol.
I am sorry you had such a horrible experience. The hairdresser had no idea about how to use Olaplex the right way. 🙁
Can you use opelex without dying it like put it in before you wash it?
It did not work on my hair at all. Everyone told me it would not work, and I listened to the articles I read instead of them. Really regretting wasting money.
Hi There
Which wholesaler or rep can I buy this from?
La Vita hairdesign
Aylesbury
Bucks
Hi,
I have never dyied my hair but I would try this product for repair and shine. Which one do I have to use?
hello, is Olaplex #2 ok to use everyday ? or will it damage my hair ? is there protein in olaplex ?
Hi, i just came upon you add about Olaplex.i am in my seventies and have been loosing my hair for about a year…and in that time i still color my hair and perm it…..i loose hand full of hair every time i comb or wash my hair. i am very healthy take the proper vitamins and exercise.. Do you think would Olaplex could help me?
Hi I just received a bottle of Olaplex , but I don’t know how and how much I can use it and how many times in the week also my hair is very long ?thanks for your answer??
Hi all,
Can I find out what is the best time to apply Olaplex? Should it actually be applied at the same time as colour treatment? i.e. does the hairdresser add the chemical to the colour treatment? or : as I have been doing for a year or so, should the Olaplex be applied as a wash out conditioner after the hair colour process has happened.
I am purchasing the product myself now as I am keen to use it between colour treatments as a hair mask.
I would think it’s best to add it to the hairdye, so bonds can be repaired immediately before the hydrogen peroxide can cap the ends as explained in the article.
Also, you can imagine that if too many bonds get broken, like when bleaching/lightening the color drastically, the hair will break and it will be too late to use Olaplex at all.
Hi Michelle and Ed,
Does Olapex have a repairing effect on hair that is damaged due to medication use? My sisters hair follicles seemed to be damaged by a period of using heart medication. Her hair fell out – and she reckons that she has only half or less of her original ‘healthy’ hair that grows left.
Any suggestions or ideas or advice?
Everything would be welcome
Thanks, Jannemieke
What is the molecular weight/mass of Bis aminopropyl?
Thanks!
Is this offer of information still going on? I have a few questions but can’t seem to find an answer(If not, if you could please point me to the right direction I’d be semi-eternally grateful):
I can see there’s a base in the reaction. Does it come from the treatment done (eg bleaching or relaxation…) or is it from a molecule in the product? Does that mean Olaplex is less effective when used on its own than with a hair-texture changing treatment?
It bonds to sulfure in a double Michael addition (thanks for the reaction btw, it helped a lot!). Do you think that by adding any product with sulfure, such as some MSM, it could do a side reaction and decrease the efficiency of olaplex? My first answer would be no, as MSM doesn’t have any S-H bond. But I wonder if I’m missing something…
Lastly: I understand that it isn’t a protein or hydrating treatment. Has anybody thought of adding pure olaplex #1 into a deep conditioning mask or in a conditioner in order to have a well rounded, repairing, proteinating and hydrating treatment? Or should I stick to doing each step separately?
Thanks a lot for any answer, it is greatly appreciated!
Hi labmuffin and Ed
I’m trying to find out why some people dont do so well with olaplex and who should avoid it
It is clear there is some great effect for most people, but for myself each time I have used it and certainly on using olaplex 3 at home there is definite breakage and thinning. During the pandemic I have stopped and my hair has returned to its usual thickness. I have curly hair and I do think I had a keratin blow dry so the additive effect didnt help but that was over a year ago. I am a primary care doctor and academic fellow, and keen to understand chemically why it may not work for some people so appreciate labmuffins hypotheses but from a full disclosure point of view I need to know if damage in these cases is a possibility for not only myself but to think about it in the 1000s of cases I see a year in female hairloss with normal biochemistry results and make a recommendation
Thank you for this in depth explanation. As someone with hair that is a fried mess from over bleaching, I have been very interested in Olaplex. The Beauty Brains did a quick summary in their forum and concluded:
“I think that the patent shows a real lack of understanding regarding the entire process of coloring the hair. While coloring does damage the hair and they may be able to reform some bonds that are broken during the coloring process, hair coloring is usually not a reduction process and the purpose of the peroxide is not to reform bonds (-S-S-), but rather to bleach the natice color and polymerize the dye intermediates. In short – I would put this stuff in the category of “More useless products for which you can charge money”.” (From http://thebeautybrains.com/bbforum/index.php?p=/discussion/693/how-does-olaplex-work )
But the results look so good! I am still seriously considering splashing out $140 for a DIY kit on eBay just to get my hair looking normal again.
With respect, I don’t think the good people at The Beauty Brains took more than a quick glance at Olaplex – I think they overlooked the fact that it’s not meant to replace the peroxide but rather to repair hair damage in general (which happens to be a particular concern with hair colouring). And I hate to be all “anecdotal evidence, yay!”, but the results are really impressive – the reduction in hair breakage after extreme bleaching isn’t really something that the placebo effect alone can explain.
Great, thank you! Something didn’t make sense to me about their explanation, and you’ve hit the nail on the head. I wouldn’t be using it with the bleach, but more as a repair job. Now I’m really tempted to try it!
Olaplex can’t replace peroxide as they have two different functionalities.
I love your blog, but I think you missed a ginormous point – Olaplex works by cross linking bonds in the hair, and I doubt there is any discernibility with it cross linking proteins in the skin. That’s the huge problem with formaldehyde – it works the exact same way.
An ingredient that isn’t used in cosmetics is being used in cosmetics and there isn’t a long history of toxicology available. It would be interesting to see what toxicology has been done with this product.
The key difference between Olaplex and formaldehyde is that Olaplex only crosslinks free thiols (SH groups), which are supposed to be crosslinked in normal hair/skin/nails (oxidation by oxygen) and only appears in a few amino acids. On the other hand, formaldehyde crosslinks nitrogens, which aren’t supposed to be crosslinked, and are in every single amino acid, and DNA to boot.
It would certainly be nice to have toxicological data!
There is an MSDS for the raw material and it doesn’t look promising.
By raw material, do you mean the bisaminopropyl diglycol dimaleate, or the chemicals used to synthesise it? Because the chemicals used to synthesise table salt are an explosive metal and an explosive, toxic gas…
And thank you for elaborating on the cross-linking mechanism! Haven’t dug down into the patent yet!
I’ve just looked at the MSDS, it conveniently leaves out information about air quality!
Olaplex does not emit fumes nor does it effect air quality in any way. As for the MSDS, everything that is required is on there.
I would be interested in seeing toxicologoy findings on Olaplex as well. Any ideas on how to find out of there are such toxicologoy studies?
I would highly suggest NOT purchasing this product through amazon, eBay, craigslist or any place similar to these. These are either diverted product, which means some greedy salon owner somewhere sold it off to make a fast buck which means 1 of 2 things –
1- you dont know how old the product could be
2- and more importantly, it may not even be the real deal. More than likely, its not. I would highly suggest you go somewhere that offers the service and do it there. (http://www.olaplex.com/ go to menu, and salon locator) PLUS, I would hate for you to shell out that money, do the service to yourself and it not be the actual product. Then you will tell everyone how terrible the product is and it didn’t do anything for your hair, when in reality you’ve not had the actual service done with the actual product.
Hope this helps.
Carolen
I am a licensed hair stylist and I just wanted to thank you for that comment. People think they can just purchase this stuff online and do it at home when they have no idea what they are doing. It’s not as simple as just mixing some stuff together, there is a reason why we pay $18k+ to get educated about these things. There is a reason why you have to have a license to do hair. I don’t know anyone that would go to Walmart and by a box cutter and perform their own plastic surgery, or try to perform their own root canals. I wish more people had some respect for the intelligence it takes to be a good hair stylist. So thank you, for your respectful and supportive comments.
Unfortunately not many people are interested in surgery and dental work as hobbies…hair on the other hand, people are willing to take the DIY route and some do so because they enjoy it as a hobby. It’s not illegal! Doctors and Dentist pay far more than 18k for their education…so Please, there is no comparison. Yes, to obtain the proper outcome hair color requires much more than just mixing things together, but believe it or not many non pros are more than capable of understanding far more than any stylist comparing themselves to a doctor would ever admit. As far as olaplex is concerned, it really is as easy as mixing a few things together. There is zero reason this product, just like all other pro products can’t be used properly by an intelligent, sensible person without a licences. Especially on their own hair. Few people fall for the diversion scare tactics anymore, by the way. I mean come on stores like target sell HUGE, UNLIMITED quantities of pro products in their stores and online. You can’t honestly believe these are diverted products, no intelligent person can! Olaplex will be NO DIFFERENT! In the end it’s always about the almighty $$. I mean they teamed up with Loreal… that should prove my point right there.
Unfortunately not many people are interested in surgery and dental work as hobbies…hair on the other hand, people are willing to take the DIY route and some do so because they enjoy it as a hobby. It’s not illegal! Doctors and Dentist pay far more than 18k for their education…so Please, there is no comparison. Yes, to obtain the proper outcome hair color requires much more than just mixing things together, but believe it or not many non pros are more than capable of understanding far more than any stylist comparing themselves to a doctor would ever admit. As far as olaplex is concerned, it really is as easy as mixing a few things together. There is zero reason this product, just like all other pro products can’t be used properly by an intelligent, sensible person without a license. Especially on their own hair. Few people fall for the diversion scare tactics anymore, by the way. I mean come on stores like target sell HUGE, UNLIMITED quantities of pro products in their stores and online. You can’t honestly believe these are diverted products, no intelligent person can! Olaplex will be NO DIFFERENT! In the end it’s always about the almighty $$. I mean they teamed up with Loreal… that should prove my point right there.
Completely agree KMW. I did a much better job on coloring my own hair than two licensed cosmetologists did; my only point in saying that is that coloring hair isn’t brain surgery.
LOL,,,, Thank you for your comment, you nailed it down to a T.
I do all my own hair treatments, and always get great results. I also did my own Olaplex treatment, and oh my word it was super easy to do myself. I think a lot of hairdressers are riding the big $ wave right now, with B S..ing their clients into thinking it’s quite a “Technical” process, before the customers finally click they can do it themselves.
Hi, Just wanted to clarify something important you mentioned re: diversion…That does in fact happen especially with certain product lines-Paul Mitchell being one of them. I say that from personal & professional experience, & there’s plenty of scientific proof to back that up.
I agree consumers shouldn’t believe all they’re told but black market sales & diversion are definitely a huge problem, along with the FDA having no regulations that apply to hair products!! So just know that when you buy hair products from anyplace except a salon, beauty supply store or distributer you’re risking the chance it could very well be tampered with, old, fake or contaminated.
I gree,
She is one hair dresser I wouldn’t go to
Hair dressers who think they know it all are the
Ones that have to say sorry after a hair due.
Olaplex woldnt have to be created if YOU HAIR
DRESSERS didn’t screw people’s hair up.
The definition of DIVERSION or DIVERTED products is this
Any professional product that says right on the bottle, ONLY GUARANTEED WHEN PURCHASED IN A SALON. No consumer knows this unless they are hairdressers. Just as no one can say how old the product is, where it came from (black market), or if it is even the same product, ie diluted, altered, completely different. That is the reason they should buy it in a salon from a professional, so they are GUARANTEED to be what they say they are.
On Olaplex? I love it and use it on myself. Just bleached mid to length 3 times in the last 2 weeks to remove unwanted strawberry tones. Added Olaplex and even put the highest volume developer on strands that had been weaved blonde before! This is a ground breaking technology that a person cannot deny when the results are that of long light, light blonde with shine and fullness! Shocked to read what some people are saying about it’s adverse results. NONE of those seen here! love it!
I want to know, and originally found this site when I googled Olaplex vs. Continuum by DS Laboratories. The rep claimed Continuum was water soluable, where Olaplex isn’t? Weren’t you just speaking of the difference between ionic bonds and the convalent bonds they use is water soluable?
So happy I found this site!
KMW – thank you for your comments….I laughed a bit out loud but only because it’s true. Many stylists do come off as if a regular person cant mix a hair dye or apply color or in this case Olaplex. I watched about 25 Olaplex videos. I have a degree and am capable of measuring the water to the product. You saturate your hair, set a timer and add number 2 for at least 20 minutes. If my hair is damaged after, it will not be my fault. My product is arriving tonight. I ordered it on amazon because Olaplex will not sell direct. I trust that after seeing 500+ 5 star reviews that it is a safe place to buy it. So what they resold it and made money?
After reading so many negative reviews I am now a bit nervous and may only do a section of hair to be safe.
I am a natural dark blonde, gone black, lightened to red now. I have been red for one year. My hair is very thin and compromised so I do HOPE this is the magic answer. I will NOT be adding the product to my color. I use Madison Reed boxed color and I LOVE it.
I get what you’re saying. Stylist should be respected for the education they put into the trade. But realistically, this is not a trade that you can compare a degree too. You just can’t. There is a reason why anyone can purchase these products. Olaplex will become available to the general public once other similar treatments come out. Every salon product begins with a license only to sell. It takes a grade school education to mix up powders and liquids. As the science they teach you is nothing beyond Highschool chemistry.
I have doing hair for 32 years and I read and watch educational material at least an hour a night. I own over 200 education videos that I have watched many times along with practicing on a mannequin head weekly. To compare education to dentistry their education ends practically at school upon completion of their degree. With the hairdresser it just begins, after 32 years of purposeful education there is still a lot to learn.( The funny things is that women are more picky about their hair than they are with their teeth).
We don’t need to lump all hairdressers together as they are equal in experience and education because they have both passed the minimum requirements required by the state board.
Really? I d like to see your hair after you cut and Colour it yourself and then!
To say the science is not beyond highschool level is a pretty rich claim when you aren’t even in the industry to know this!
I’ve been an educator for many years and worked in Colour research and development and trust me there’s a lot of knowledge that goes into the science of coloring hair.
WOW….SERIOUSLY?!?! I doubt this product will be available to the public. There is education for cosmetologists for a reason, they are regulated by the state for a reason. There are reasons why products like this are not available to the public….reasons being to prevent them from being in the hands of someone like you, that think they can just mix up whatever they want and expect professional results.
KMW, I could not have said it any better, for EVERYTHING you mentioned are facts and something which I’ve been saying for years. The beauty industry is my life, I’ve even moved on to Aesthetics M.E., Trichology, SPU, Lashes & Perm makeup…. but I’m not complaining bc unlicenced people can purchase it, BIG DEAL, good for them! Amazon sells 1.2 & 3 but people are always turning them in or trying to convince people that it’s fake. Truth is, and this came from Olaplex’s page, these are not watered down or fake. Who in their right mind, would choose spending more cash on something they can get for less….A LOT LESS? I am not saying what I do is easy, but it sure in the heck isn’t Rocket science or Brain surgory. There’s NO comparison and a total joke that you tried to, AS IF!!!!
Hello
You are criticizing the ones that purchase products to
do their own hair, and you shouldn’t, because you say
something important and that is that beautician pay 18k
To become licensed, but then you damage hair as much if not more
than the person doing it on her own, if ones buns or over
bleach her own hair at least she is not paying for that to one that claims
To know it all, only because she paid 18k to because a hair dresser.
You hair dressers should remember that continues education in this
Profession is a MUST! but you are so bad that you don’t make enough money to go to seminars and new products teachings etc. then have the nerves of doing something you don’t know, screw up and then say: I am sorry. If you were so good you wouldn’t be in the Internet looking for info
You would’ve had this class already and actually learned something.
Yes no one would do their own root canal but a Dentist pays $500k to become one, and when they screws up at least there is a settlement to get a better tooth but when you screw up, their is nothing but a long
Wait for the little hair to grow plus the money spent on you..
Hello the reason i need an advice is that the salon i go too doesn’t even know olaplex and i have watch many videos i we respect your integrity of your profession but with all due respect i go to salon every 4 weeks to cover roots my questions is can i use step 1 and 2 at my home as a treatment than go do my highlights the following week plz help.
As a manager of a Beauty Supply for professionals I am so glad this was brought up.Please don’t purchase on Amazon EBay etc.Not only because it could be bogus but because this is product IMO that is best used by the hands of a licensed professional. Even tho it isn’t a chemical it is a new innovation and personally I am blown away by Olaplex.
In response to KMW.No we don’t pay what a doctor or dentist does for schooling.Apples to Oranges.Any who as a licensed hair stylist for 33 yrs I take my profession very seriously and take on going education trade shows and. numerous certifications.I think some people totally dismiss our profession and by the way we can look at someone and know right away if someone’s hair was done by a professional or not.
This is not helpful advice, and it does not help people who cannot visit salons and must do their own hair, for different reasons that are their own business. “Licensed professionals” who are “color experts” and “who went to beauty school” have burnt my hair off at bald spots on my scalp with bleach by putting it under the dryer, to shave off 20 minutes and squeeze more appointments in, even though I told them it has always lifted fine without the dryer.
I gree with you on big scale my friend!!
Well set.
I usually respect their opinion but in this case it works although you can’t expect miracles. Also, they fail to address previous damage or environmental/styling/daily damage that occurs every day. Kind of like Michelle said damage in general. I can say from experience they are flat out wrong.
HI. I was wondering if you could tell me if Olaplex and cleansing conditioners would be incompatible? I had the just the treatment, no color or bleaching etc, and since then my hair doesnt feel as smooth and soft after “shampooing” it with my normal WEN cleansing conditioner.
Unfortunately I don’t have much experience with cleansing conditioners – have you tried using a normal shampoo and conditioner a couple of times and seeing if it changes how your hair feels?
As a cosmetologist who uses Olaplex in my salon with fabulous results I will tell you it’s the Wen causing any problems. I have seen hair transformed with Olaplex and I use it on myself and can tell you it’s a miracle product for my industry.
http://www.kisforkinky.com/wen-cleansing-conditioner-causes-hair-loss-wen-hair-care-lawsuit/
Today my stylist was saying she has had 3 or 4 people that had hair loss, breakage, or bad results from wen. I’ve never tried it but didn’t think it was that harmful nor great.
Unfortunately I would not recommend using a “cleansing conditioner”. Since you are using this product I would recommend using a clarifying shampoo, prior to doing a stand alone treatment. When using a product such as that one, it contains a good deal of silicones and even wax. When this type of buildup occurs, Olaplex is unable to penetrate the hair.
You should use professional products when using Olaplex. You might want to see this
http://www.usatoday.com/story/money/business/2015/04/02/wen-hair-conditioner-lawsuit/70822018/
Did you hear about the huge recall on Wen products and their several law suits. I was shocked but worth looking into, maybe it’s their products.
I have a client who has been using Wen for years! She loves it… But I have noticed her color processing time has been longer and longer. The light came on when I used Olaplex in her high lift color ( hilites ) and then followed up with 2 treatment for 15 minutes knowing she is going to love the way her hair felt. NOT it actually felt worse. I went on Olplex FB page and asked why this happened? Popular response was Wen needs to be clarified ex Maibu treatment, before color and Olaplex. I’m guessing some cleasing conditioners BUILD UP so much that the hair can’t receive the proper color and Olaplex can’t work it’s majic. So I believe that a clarifying treatment should be done on a regular basis. PS I was also informed that Wen has pending lawsuits against them in UK and United States. I briefly researched this and found some people blame Wen for Alopecia. Don’t know if it’s true but if the scalp is coated and not clarified how can root produce healthy hair? Just a logical observation on my part.
My hair was healthy I had it highlighted foiled left on about 40 min nothing abnormally long. Opalex treatment one and 2 prior to bleaching and mixed in bleach. Then opalex 2 after bleaching at salon.Hair was ok when I left salon highlights effect was good no orange or yellowing. Blonde amd light brown i was pleased. Then after first shampoo and condition as I always have.my hair was very fuzzy dry sticky. My natural curls structure was no more and it was very brittle but I kept condition ing and after a week it was started to get some shine getting slightly better. Then I put on step 3 opalex used it generously as it said on bottle left it in for about 4 hours washed out and tried to condition but it will not even soak in is sticky doesn’t matter what kind I keep trying to deep condition and it’s not helping. My hair is stripped and the highlights are orange and yellow. My hair has never been worst. I have fine hair that grows slowly and I’m 47 yrs old. My hair seems to be getting worst more dry nd straw like each day. How can I remove this product from my hair. I’m absolutely devastated. I can’t lose my hair. Please help I emailed opalex no reply.this stuff can have bad results. People need to know that step 3 is not a conditioner and can harm and there needs to be a warning not to over use and leave in. My hair was fried from the first wash and condition. It’s like my shampoo and or conditioner were not compatibile with opalex. I certainly had highlights before never this result. And the orange and yellow ing after step 3. My hair is stripped a direct result after step 3 . I’m also thinking of contacting a lawyer. This does not wor k with everyone and every product . It has done the exact opposite of what I was told. Can anyone help me fix this mess? Please this is a nightmare.
You need to have used Olapex 2 right after rinsing out the bleach mix (with Olaplex 1). Or did you directly went to the salon with towel dry hair after rinsing? If not there was the mistake. It also depends on the shampoo you use. See the Wen issue.
As I am no professional but have read a lot about Olaplex lately I am wondering why you used 1 and 2 and then 1 again in the bleach but forget to use 2 like you are supposed to?
@Trina B – pls contact me via e-mail if you managed to do something about it because I experienced exactly the same problems like you. This is really a nightmare and I’d like to sue them as well. My hair was thick, healthy, shiny, natural…and now, after using Olaplex1,2 and 3 there is no volume, it’s stripped with orange, fuzzy at ends, oily, thinner and thinner every day…I can’t stop crying, looking at the mirror and touching straw on my head:-(((! Thanks a lot
Trina B and Ivana I experienced the same disastrous results. Just using the standalone Olaplex 3 without any colour or bleach and my hair is absolutely ruined! It went from being shiny and soft with lots of volume to brittle, fragile, coated feeling and thin. I am completely lost at what to do without having to shave it all off 🙁
Great article! A few questions:
Because this ingredient slows down the process of lighteners, the directions say to increase the volume of developer to compensate. So the question is if left on for the same time, is there an increase of damage from having to bump up the volume of developer from say 20 vol. to 30 vol.?
You allude to the hair being harder to repair after this treatment has been used (just a theory). To me that sounds right because other treatments will be “looking” for those free thiols have been capped and will not to attach. So how would this effect treatments such as Cysteic Acid Treatments, Ceramides, or even Biotin?
Also a follow up, because that “link” Olaplex creates is water soluble, what would be factors within the hair that would define how long it stays?
Re: Increase in damage – I don’t know, but from people’s experiences the Olaplex seems to compensate for any extra damage the higher peroxide content causes!
Re: Other treatments – I haven’t researched those treatments in depth, but from my brief Googling it sounds like they do interact with free thiols, and hence won’t work on the thiols in hair that have been treated with Olaplex. But in damaged hair, not all of the disulfide bonds are broken, so those treatments should still work with any disulfide bridges that have broken after the Olaplex treatment.
Re: Olaplex link – I’m not sure if it’s water-soluble – there are some ionic bonds which are water-insoluble and can stay around for a long time (e.g. in marble). And it’s hard to say for sure what will make the bonds break faster without any direct studies. But there are a few things which will make all chemical bonds break faster – any input of energy (sunlight, heat, physical movement). And if it’s water soluble, then obviously washing hair less will help.
Hi Michelle,
Thank you very much for your post. As chemist I’ve been thinking about the effect of the ionic bond between maleates moieties and the linker.
After a probable breakage of the ionic bond, what do you think about a possible reaction of the carboxylic acids forming esters or amides?
I’m concerned about a percentage of keratin cross-linking using flat irons after a Olaplex treatment.
Thank you very much again, and congratulations for the detailed post.
I’m a stylist and have been using Olaplex. I have noticed a distinctly better texture to the hair after lifting, and I use Olaplex exclusively in my bleach processes (Balayage and foil applications, 40 volume and Platine, which is an accelerated bleach for hair painting.) I do dislike how it makes the process so much slower, and I do feel I get less lift with it in my formula, no matter how I adjust the volume of developer, and how long I leave it on. It’s like bleach has a time-limit, and once it’s reached it, it just stops working. Sometimes I find myself skipping the Olaplex and treating after or before instead.
I really appreciate the article, because I have had a hard time explaining how it works, and understanding it myself. This demystifies it for the most part!
I’m curious to see if you can perm with it now that you mentioned it. Are you suggesting rolling a perm as usual, then using Olaplex to saturate, process for a set amount of time, and use Step 2 as the neutralizer? I have been wanting to see if I could do a Curlformers perm on a doll head, and that might be a good combo.
I’d love to see a follow-up on your questions regarding covalent bonds. I didn’t realize that the effects of repair were so short-lived. In my head, repaired is repaired, not an illusion. That makes Step 3 (retail for clients) really important, as it is prolonging the effects.
There are instructions for using Olaplex with perms. I am pretty sure you saturate the hair with Olaplex after risnsing and before neutralizing.
All of the directions are available on our free App for Apple and Android phones, Olaplex.com and Olaplex users group forum on Facebook.
-Ab
Senior Educator
Olaplex LLC
Ola, fantasticas as perguntas e respostas sobre OLAPLEX, sou Cabeleireiro No Brasil e temos muito problemas com LOIROS, principalmente devido ao uso de FORMOLDEIDO, ACABEI DE APRENDER MUITO com este forum , com certeza eu gostaria de ser um educador OLAPLEX no Brasil para que seja melhor aproveitado.
Apply Neutralizer To Each Rod, Time For 5 Minutes. Rinse Rods And Towel Blot. Apply OLAPLEX Over Each Rod, Set A Timer For 5 Minutes. At The End Of The First 5 Minutes, Reapply OLAPLEX Over Each Rod And Time For An Additional 5 Minutes. Remove Rods And Rinse Thoroughly.
Hi,
I had platinum blonde hair that had a regrowth of 1.5cm. I wanted the lady to make my regrowth look softer, she used bleach on my hair with Olaplex. I really do not know why she used bleach in the first place. But my hair snapped off. I left the salon with half my hair left. What could she have done wrong for this to happen? She said that she overlapped on to my blonde with the bleach.
Ohh my goshhh that sound so familiar!!!
Why they pay 18k If they are going to
Suck at doing hair? and worst they dare to even temper
With your hair instead of admitting that what
You asked for its out of their $18k knowlage.
Congratulations. Beautiful explanation of a possible mechanism of action of Olaplex.
Can you explain how the hair straightening products based glyoxylic acid works? I don’t think that the glyoxylic acid can act on bridges sulfur at pH around 1.5, yet it work very well and for long time ( 3-4 months) ). This kind of hair straightener now represents about 90% of the market for hair straightener products .
Este produto Glioxilico é uma febre no BRASIL , usado em ESCOVAS PROGRESSIVAS PARA ALISAR OS FIOS, mas tambem vem causando serios danos aos cabelos bem como queda silenciosa e um profundo apodrecimento do cabelo, que não se consegue recuperar.
Thank you for writing this post Michelle! I just had Olaplex on my hair and want to write about it but even my colourist couldn’t explain exactly how it worked. Can I reference part of your post in mine?
Of course! 🙂
What do you think of the safety of the chemicals in Olaplex? I run an organic hair salon and use the most natural products that I can get my hands on to limit the amount of chemical exposure to myself and my clients.
I’m curious if Olaplex could be something I could incorporate into my services but I don’t see much info on the toxicity and or lack thereof of bis-aminopropyl diglycol dimaleate.
Thanks for the article!
Hi Julie, I myself run a mainly organic salon. Olaplex is non-toxic but is still a chemical. Most products labeled “organic” are not truly organic. Nothing can be considered organic unless its pulled straight from he ground and refrigerated. I have been using Olaplex for well over a year, and tested it until the launch to help with development. If you have any questions, you may email me at Ab@Olaplex.com.
Ab, everything is a chemical. Water is a chemical. Oxygen is a chemical.
“Chemical” does not equal “bad,” any more than “organic” equals “good.” After all, poison ivy growing in the woods and bee venom are “organic.”
I’ll used it and it with my hair hard and I wasn’t even hardly able to put a brush through it. I was wondering why
@Jordan hill-
Olaplex is only capable of multiplying disulfide bonds. If you are having issues with brushing, you will need to address protein and moisture as these are entirely separate issues. Olaplex works internally. Also, feel free to email me.
Do you know by any chance how long the olaplex effect last? it made my hair looking horrible and really fizzy !
Thanks
Cristina
The same thing is happening with my hair and I would like to know how long this lasts. It’s terrible!,
Yes I agree it’s horribe.
It’s horrible. I want my old hair back. How long does this last?
Works great with perms and you can wash and condition immediately!
Hi Michelle. Thank you so very much for this article.
You know there is a lot af different products claming to do the same. I was wondering if you could do the same with these : Colorphlex and Niophlex, also known as Eslabondexx. I would love to know the difference between the products from a “neutral” person.
Thank you again.
The American Board of Certified Haircolorists, or ABCH, recently posted an article comparing Olaplex.
http://myemail.constantcontact.com/OLAPLEX-Tested-Again.html?soid=1102356594365&aid=RGJVd8Idfn4&mc_cid=f035c57cba&mc_eid=3b67fcf90b
I have heard great things about this product, but I heard great things about Japanese straightening and keratin straightening the first year as well . Those products went south for different reasons. I am curious to see how Olaplex does over time.
I’m curious too! I guess we’ll just have to wait it out and see…
Olaplex states that the percentages of active ingredient are 100% in No. 1, 15% in No. 2, and 12.5% in No. 3. No. 1 contains only water and bis-aminopropyl diglycol dimaleate. No. 2 and No. 3 contain additional ingredients, and have the exact same list of ingredients.
Olaplex’s directions for a standalone treatment include saturating dry hair with a mix of 15% No. 1 and 85% water, i.e. 1/2 oz (15 ml) of No. 1 and 3 oz (90 ml) of water, or 1/4 oz No. 1 and 1.5 oz water, etc.
Scientifically, there’s no need for a 3-step system. One could simply apply No. 1 (diluted to 15%) or No. 2 (already 15% in bottle) or No. 3 (already 12.5% in bottle) individually and repeatedly. In fact, Olaplex states that it’s fine to use each step individually and repeatedly if you run out of a certain step.
For licensed professionals (and students), a traveling stylist intro kit costs $65 (USD) and contains 3.3 fl oz (100 ml) bottles of one bottle of No. 1 and two bottles of No. 3. The 3.3 fl oz bottle of No. 3 is sold separately and costs $14 (USD). For home consumers, the current market prices are $90-$110 (USD) for the intro kit and $20-$30 for the No. 3 sold separately.
Olaplex states that each kit contains up to 15 applications. 100 ml bottle of No. 1 divided by 15 applications = using about 7 ml (about 0.24 fl oz) of No. 1 per treatment.
Oops! Correction: The traveling stylist intro kit contains one bottle of No. 1 and two bottles of No. 2.
This might seem like a dumb question, just wondering, do you have to use steps one and two to use step 3– the client take home. I’d imagine using all three would be ideal, would one see any result from using step 3 alone?
Thank you so much this cleared up so many questions for me. Hope will see a olaplex 2.0 in the near future lol ;).
I guess my questions would be after doing a ton of research based on chemistry and Im not a chemist; nor can I spell it
is it then just a band-aid and not permanent
can it build up on the hair until it wont work any more
(im not a scientist so this is the only way I know how to say it. and I know im not supposed to be a scientist LOL Im a damn hairdresser anyway LOL!)
if you stop using it does your hair go back to the way it was before you started using it.
coiuld it work instead of neutralizer on a perm and maybe make a perm feel more like real curly hair instead of unnatural curl intil it grows out for 10 months.
these are questions that I cant seem to find answers for. I believe now that I understand the chemistry behind it. Kinda like someone who only knows enough Spanish not to go to jail.
You rock girl!
I’m not sure about this – it’s certainly more permanent than most of the other treatments available (e.g. keratin).
Again, I’m not sure about this! I think after a few more months we’ll find out from people who have had Olaplex done.
It should take a while before it reverts (again, more permanent than keratin)
It should work as neutraliser – I don’t know how it’ll feel unfortunately! I believe some people have had experience using it this way – they might have a better answer!
From personal experience having done MANY olaplex treatments to my hair standalone and with bleach/lightener I can say yes it is permanent but I would maybe best describe it like a perm. Like months later my hair is still less damaged, but I think over time even just washing and exposure without additional standalone treatments like I did before, it is not as evident it my hair, but it is still there. Just like permanent color fades, but it isn’t gone. I can’t speak to buildUP per say but it does build upon previous treatments because my hair had that yucky mushy soft feeling when it was wet and it went away after multiple treatments. I also stopped having super easy breakage and that hasn’t come back in that hair either. I have since been trying to get a medical condition under control so hopefully my fine hair won’t be breaking off anymore, but I’m sticking with the olaplex.
Hi Michelle, I have a question.
Explained action of Olaplex is proper when disulfide bonds are broken during straightening or perming work and free thiols are available.
But during bleaching or colouring, in oxidative environment, disulfide bonds are broken via formation of oxides and sulfonates are formed. See:
http://journal.scconline.org//pdf/cc1971/cc022n06/p00339-p00348.pdf
this article is old but still cited and verified.
Are there other mechanisms of action in Olaplex patents explaining in which way, in these situations, bonds could be reconnected?
I can’t remember if this was in the patent or from a video interview with the patent holders, but I think they explained that the Olaplex reaction happens faster than the sulfonate formation reaction.
Olaplex is amazing. As someone who turned prematurely grey at 18, coloring my quick growing hair every three-fourths weeks, plus modeling, my hair works harder than I did. Thank God for Jennifer at http://www.dipietrotodd.com/index.php
Has anyone tried the new alternative product yet? It’s called Continuum and it’s made by DSLaboratories. It actually is based on 3 molecules as opposed to 1- no need to bump up developer or add extra processing time. It’s silicone free. I can’t and won’t do hair without it. As a stand alone treatment is my favorite. I’ve never been more impressed or obsessed with a treatment!!!!
What are the 3 molecules of the Continuum? In the DS Laboratories I don’t found the ingredients of their products. I hope it is not other … ghost … molecules (lately one sees many outstanding).
I’ve heard of it but have yet to read any reviews or side by side comparisons to olaplex. When my personal stash of olaplex is gone I’d like to get some. Do you find it is better?
Has any one had a reaction with their nails? Mine are splitting.
Karyl
Has anybody had trouble with pealing nails from using olaplex
Are you able to explain to me what the difference is between Olaplex and Cureplex – aside from the price?
This is actually the first I’ve heard of Cureplex. I’ll have to do more research!
I would also like to hear your comments on the difference between Olaplex and Cureplex. I am told they are essentially the same thing, just different brands. I am a little sceptical… Thanks
Most in Australia seem to be using cureplex. Been told it the same. Would love to know the truth.
Cureplex is definitely NOT the same as Olaplex.
Do some research into the ingredients lists of both and educate yourself. There’s an enormous difference between the ingredients in Cureplex and the ingredients in Olaplex,.
From now on, I will only go to salons that use Olaplex.
The reason a lot of salons in Australia are using Cureplex instead of Olaplex is because Cureplex is cheaper – and so, in my opinion, are its ingredients.
The ingredients are what make an enormous difference between Olaplex and Cureplex.
I personally don’t like Cureplex, but there are plenty who do.
My own Cureplex experience (as a customer) is that the #2 at home treatment leaves my hair lank and horribly greasy, no matter how little of it I use – and I confined it to the very ends of my hair, but it still grabbed a hold and weighed it down with grease by the time it was dry. Yuck.
Also, when the bleach was being rinsed out of my hair, it didn’t feel any different to the crunchy, dry way that freshly bleached hair normally feels.
I’ve studied the ingredients in both. Cureplex uses way too much Cetearyl Alcohol (a fatty, waxy, alcohol) and I’m going to guess they also add way too much of their silicone blend – which is what all their claims are pinned on – and which the manufacturer, Croda, describes as ‘extreme heat protection’.
There’s ‘extreme heat protection’ and there’s too much of a good thing leading to lank, slimy, greasy hair.
My next appointment to get my roots done will be with a salon that only uses Olaplex.
Olaplex’s ingredients are entirely different to Cureplex – in fact, there’s no comparison – and I noticed that it also incorporates some form of amino acids (always a good thing). Although I’m not a chemist, I can see how Olaplex would be far more effective at healing and repairing hair damage where Cureplex is just a ‘conditioner’.
Also with Olaplex, from my understanding, you don’t absolutely need to purchase a product to use at home, whereas with Cureplex, you do so for me, Olaplex will be the way to go.
I just had a Olaplex treatment. I’m in awe with my hair!! My hair is very course, thick, and I have it highlighted. Aside from all of the above my hair was in okay shape. Until we had a soft water system installed in our home. It literally fried my hair! My hair felt like a brillo pad, straw, and I thought I would have to cut it all off. After just one treatment my hair feels like silk! I don’t know how long it will last but I was advised to do another treatment in a week since my hair was so damaged. My question….how long does this treatment actually last? I hope it helps me until my hair grows out and goes back to being healthy!
Hair can be damaged from chemical services, mechanical such as brushing and especially thermal such as blow dryers, flat irons and curling irons. By using the Olaplex treatment, you link broken disulfide bonds therefore restoring the strength, structure and integrity to the hair
. It lowers the porosity of the hair and increases elasticity in the hair. Its an overall reparative treatment! You can do the treatment before your color to strengthen the hair. This is a great pretreatment for working on compromised hair or before a drastic color change. It is not a protein, you cannot do any damage by using it too much. The more FREQUENTLY you do the treatment and the LONGER you leave the treatment on – the more effective. You can also follow any color service with the same treatment just to restructure the hair after coloring it.
Tried this product and did not have work one bit!
I’ve had 2 Olaplex treatments done, 6 weeks apart. My hair is thick, bleached, shoulder length, and very dry. Olaplex did nothing for my hair either.
Michelle ! boa tarde, vinha buscando varias respostas a este novo produto e acabei encontrando aqui, muito obrigado apendi muito sobre OLAPLEX.
I would also like to know if cureplex is the same.
How long does an Olaplex treatment last?
If you have extra step 3 left over can you reapply it in another 4-7 days or is it redundant?
I used the stand alone treatment, and my name is Tina not tuna lol
I just wanted to share my experience with Olaplex. I am a licensed cosmetologist and after seeing rave reviews I wanted to try this product on my platinum blond waste length hair. But what came afterwards was a horrifying experience! My hair broke off in big clumps and now months later I am still experiencing breakage along the same breakage area line. I have done my own hair for years and have never had this horrid experience happen. I have half as much hair as I use to and all I can do now is wear it slicked back with clip in extensions to hide the breakage and thinness. So of course I was devastated and I re-read the directions making sure I did not make a mixing mistake and I did not. So I searched out to find a represenitive to tell about my horror story! I was given a email address for a company represenitive at which time I sent her this email….
Me:
Hi Sarah,
I was given your email address to share my experience with olaplex. I have been platinum blond for years. I am a licensed cosmetologist. I used olaplex this last time I did my regrowth and my hair broke off. And not like a little breakage this is massive breakage in my crown and Mohawk area. My hair is maybe a inch now in those areas and I have hair almost to my butt! This is extremely bad for business when your know for blonding. I now must ware my hair back and hair sprayed down. My hair is extremely dry and every time I wash my hair more breaks off! I am really trying not to have a melt down over my hair damage. I just wanted to share my experience and voice my concerns.
I used 1 oz Schwartzkopf bleaching powder with 1/4 oz Olapex and 40 volume developer because instructions say to boost the developer one level higher than my usual 30V and I do not use heat. This is a on the scalp application. I let it process about 40 min. I have never had problems lightening my hair before and having breakage. I’m gonna have to cut my hair to a short pixie cut now! My hair is to my butt! Every time I wash my hair more and more breaks off!!! This is even with me washing in cold water and letting my hair air dry with no heat! Honestly I’m devastated!
Olapex Represenative: I apologize about such a terrible experience you have had with your hair.
I copied this from the FAQS sheet to show the directions for on scalp lightener. Due to client’s scalp sensitivity we do not suggest using anything higher than 20 volume. Using a higher volume is done at your discretion.
Did someone do your hair for you or did you do your on scalp application yourself?
Was cotton applied at the line of demarcation to avoid any over lapping?
40 minutes is a long amount of time to leave 40 volume(30 vol with Olaplex) on and I feel if there was any overlapping, even the slightest amount, the high volume could have caused breakage on your already lifted hair.
As for lightener, it may also be added directly in as well. By doing so, you are actually able to rebuild bonds as they are being broken during the lightener process. When you lighter hair, you lose about 50% of the bonds. The next time you lighten, you lose about 50% of the 50% leaving you with 25%. It gets exponentially worse from there. By adding Olaplex to your lightener, you are actually able to rebuild at the same time allowing you to push hair farther so there are more bonds to be broken. So instead of initial 50% or so, your losing 10% give or take dependent on hair type etc leaving you with the ability to push hair farther. I can get into specifics of mixing etc if you like as well, this is just a generalized explanation.
————————————————-
So with all of this emailing I concluded that there claims of “NEVER BREAKING HAIR AGAIN” are false and misleading. Also if this product is a safe guard from breakage why would it matter if you over lapped product? I always slightly feather out my product so there is no line of demarcation or missed areas. Using 30v on the scalp for up to 40 min is what I have done on my own hair for years and never had this reaction. In her response she states that “When you lighter hair, you lose about 50% of the bonds. The next time you lighten, you lose about 50% of the 50% leaving you with 25%. It gets exponentially worse from there.” Ok so if someone is not overlapping like she stressed to me could be a user malfunction then why even use the Olaplex, cause then you would not have the 50% of the 50% further damaged. Also she stated “By adding Olaplex to your lightener, you are actually able to rebuild at the same time allowing you to push hair farther so there are more bonds to be broken”. Ok I’m assuming she meant, there are no more bonds to be broken. But anyways, obviously there are lots of contradictions in this conversation, she is saying that limits can be pushed but don’t push them. Also I got to thinking, if you use a level higher volume developer like the instructions tell you to use (might I add the instructions said nothing about on the scalp application or that 40 vol would remain at the same strength if used so don’t bump up 30 to 40) then you would insure the same amount of damage as the higher volume developer and therefor if you used your regular formula without the higher vol developer you would not subject your hair to the increased amount of damage. The olaplex claims to be a safe guard for damage and allows the cosmetologist to push the hair farther are dishonest and could actually put a professional in a extremely horrible situation. If you just use products accordingly and take time to let them process there is no need for a advertised safe guard that is actually not really a safeguard. Also I must add that after consulting with a Schwartzkopf educator they do not recommend adding any additives to there products.
I also had my mid length blonde hair break of into 3 inch pieces from the use of Olaplex with bleach. I have gone from red, to honey, to dark brunette to honey all with minimal damage until my stylist used Olaplex. I firmly believe it was from this product.
I too experienced horrifying results. My hair burned off in clumps and just so happened to be at the front of my hair line. I lost everything on the left side of my hair about an nch back from the hairline. I also lost the majority of my bangs. Some ended up being a half an inch long in some areas. It is so bad I can’t cover it with extensions either. I am miserable looking at it and trying to figure out how to hide it every day. It literally brings me to tears. It felt like a brillow pad after each application and has cost me hundreds in hair repair nutrients and conditioners. It makes me SICK!!!! Devastating.
I definitely know how you feel, here’s an Attorney you can Contact Janet Varnell 352-753-8600 she is handling the “Wen” hair loss product case.
Here we go again with “Olaplex” not taking responsibility for their product!! Same old answers putting blame on the stylist or the consumers!!!
This stuff fried my hair also. I have not gone to work for 2 days . Obsessing on how to repair this damage. Condition ing but nothinges helps and my hair is getting worst. OMG this could not of happened at a worst time in my life. Stylist need to know this stuff can do this.
Can I leave the product on my hair for a couple hours?
It has really healed my hair. It was so fried it was literally shredding apart.
I have a client who purchased it on amazon and the colour of the #1 was completly off!!!!! It definetly was not the true product as I have in the salon!!!!! BUYERS BE AWARE
Fantastic read. I wish I could send you some Olaplex for a follow up post. 🙂 Thanks!
Love the way you put things together!
Thank you soooo much for doing this. It is really helpful!
I have a request for you. Do you know about hairprint? http://www.myhairprint.com
It is a mistery to me how does “restore one’s natural color” as they claim:
“When you replenish the hair’s natural pigments, it reverses gray hair.” “think of it as a Hair Healing System that just happens to reverse gray hair to its natural color” “Hairprint creates a process whereby the natural pigment in your hair called eumelanin is recreated in the hair shaft. Eumelanin is arranged in the hair in a pattern unique to each person”
And its ingredients are pretty simple:
“The ingredients are water, baking soda, mucuna pruriens, sodium carbonate, carbomer (a thickener), hydrogen peroxide, diatomaceous earth, manganese gluconate, and ferrous gluconate.”
“Hairprint is not a dye. The same product can restore gray hair from very light brown all the way to the darkest black. Your hair will determine your color. It will look like your hair color because it is”
If you could give us your input over this, I would be delighted.
This looks super cool! Thanks for bringing it to my attention, I’ll definitely need to read up on it!
You are ver welcome!!!
Found this interesting:
http://www.google.com/patents/US20020166182
I wondered if any medications could cause a bad chemical reaction with the Olaplex? When I used it, my hair smelled STRONGLY like isopropyl alcohol for a few days!! I got the first two steps done at my very qualified salon. I took home step three and am afraid to use it again. I tried it two more times-no alcohol smell this time, but I’m sure my hair was healthier before all this. Help! (And thx)
Im not sold by Oplaplex either and im a professional distributor of hair products and own 3 pro salons. One thing i cant get to grips with is the sheer marketing prowess Olaplex have invested in, it seems they manufactured using very cheap basic packaging and invested the rest into a huge marketing campaign.
Also i can see on any forum that speaks bad or questions its legitimacy, there is an office of paid marketeers with multiple accounts jumping on to defend their product. Or as we see above, telling us we cant buy it online and must only buy it from their representatives, that are obviously being paid to make videos all over youtube.
I have tested olaplex and a few of the other similar brands, what did i notice? it did nothing countless other hair treatment or keratin treatment wouldn’t do. Except it has been marketed for a specific niche to fix a specific problem, again clever marketing.
A good colorist wouldnt need it in my opinion.
This actually happened to me on the Rainbow Haircolour FB board. I’ve never experienced anything like it. It was a major flamefest, including several personal attacks, people trying to twist my questions, calling me a moron, telling me I’m stupid and acting like three year old, berating me endlessly and various other manipulative verbal attacks for being merely skeptical. My “crime”? Asking if there were any unbiased, scientific peer reviewed research for this product. A truly bizarre experience. Clearly, the product isn’t good enough to stand up to simple, logical, valid questions. Let’s look at some simple logic. If it permanently repaired damaged hair, why would you need to continue using it between chemical services? How could it make it “better and better” ( company marketing claim) if it’s already permanently repaired? Contradictory, isnt it? Under the circumstances, I won’t be using this product.
It’s not a contradiction. The chemical sevices keep breaking bonds. Olaplex doesn’t promise to prevent a bond from ever breaking again it simply repairs the ones that you just broke with the chemical service. But if you keep breaking them what do you expect to get but broken bonds after broken bonds.
Hi! Wow its so cool to find such a thorough explanation about a hair product! The information we receive as hairdressers isn’t as informative, its usually sounds a bit magical and vague. I would be really interested to see how you would compare the differant products in this new category of “bond multipliers”. I have been using Cureplex in the salon and have been happy with the results so far, but I can’t see the same ingredient used in Olaplex, and product reps are far from objective about the differences between the products. I will have so much reading to catch up on your other posts now, yay!! Thanks you!
I was given an OlaPlex treatment at the end of July and again at the end of August to treat hair over processed with a perm and color and hilights and the results were beyond spectacular! I never dreamed my hair could look that beautiful, with or without all the processing I’ve been doing all these years. So I informed my salon that I wanted to add OlaPlex to all my future treatments at which time I was informed that my salon would be going with a product called Continuum and would not be using OlaPlex. To counteract my wild reaction, they offered me a Free full treatment of Continuum. Well, I hated it. It softened my thin hair to the point that it had no body, no bounce and was actually flyaway. I don’t want to leave my salon because I’ve been with them about 13 years but I can’t see giving up OlaPlex either. Also, I live in Westchester, NY and can’t find another salon that offers OlaPlex.
Any advice?
I have a salon in Oregon and was pushed by a rep to use Continuum also, as a swap out. I didn’t like the first treatmwnt I performed either. I tried it a couple other times, complimentary and people either said they really couldn’t tell any difference and one said it made her hair weird and color washed out sooner than seemed normal.
I’m staying with olaplex, because I know it works!
check salons in rockland county ny…….everyone here has it…..im just not sure how i feel about the product.
Thank you.
I was suggested to do the Olaplex treatment at the salon I normally go to and was sold to me as a miracle hair repair. I wasn’t aware my hair needed repair but I was advised to do it anyway.
the end result wasn’t good. My hair have lost all volume and even oif I blow dry it with a round brush or roll or applying a volumiser it won’t take it. Plus my hair now looks really fizzy and brittle which didn’t before. I really hope my hair will be back to normal soon. I’ve done the treatment 22nd September but still is looking bad. I wish I never had it done! and if anyone has any solutions or recomendation on how to get my lovely hair back please let me know!!
Thanks
Cris
Hi,
I have virgin hair (at 24 years old) I recently had a hairdresser burn it with curling tongs! Which was devastating as I don’t style with heat at home and have never dyed it.
After trying numerous things to get my hair back to it’s beautiful, shiny, non-breakage self OLAPLEX was the only things that has helped. It’s soft, shiny and the split ends have now disappeared.
I have very sensitive skin and I’ve had no reaction to this. I don’t use no.3 at home I’ve just had two in-salon treatments in the last two months.
Please give it a go, my hair is manageable and has body again.
Brazilian blowout has a split end corrector treatment, which could be great for that!
As a scientist I have to say that this molecule is my new BFF. I haven’t done it with bleach, but I have done it immediately after as a stand alone treatment. My hair is by no means healthy yet, but this kept my hair from turning into a ball of protein mush. I will continue to do treatments until my hair is repaired to my liking, and then I will do them regularly thereafter to retain results.
BTW, “anecdotal evidence,” also known as qualitative evidence, is completely acceptable in scientific research. Researcher just can’t claim as much of a universality to their findings- just saying…
Re: WHY ARE THERE IONIC BONDS IN THE STRUCTURE?
I think part of the product design is that the repair is meant to be impermanent. Although people do have repeated treatments on their hair that would continually cause damage, as well as new hair growth that gets treated, a permanent solution would not require take home treatments and as frequent reapplication. The water soluble nature of the linker means it will wash out, resulting in multiple applications of the product required to maintain the hair texture and thus greater sales/revenue. Or maybe that’s too cynical? It seems pretty clever from a product development point of view, however.
These are all logical, valid points and not the least bit cynical. Savvy consumerism is the intelligent way to go.
Hi Michelle,
thank you very much for this valid information about the chemical background. I reached out to my german audience …
Best regards
Marita
Olaplex hair treatment was used on my hair by my hairdresser and unfortunately it is now in the worst condition it has ever been! If you are thinking of having it don’t! My hair was only done 1 week ago and everyday since then it feels greasy, dirty, very fine. It is also full on split ends and is snapping also in the middle of the shaft. It will not style and when you run your hand through it the hair sticks up as though it is greasy. Oh I am so upset about it, I have always looked after my hair, if I had researched it first I would not have had it used on my, but I trusted my hairdresser of 5 years! Now I have another hairdresser trying to find a rescue remedy for me.
Please let me know, did you get your hair sorted? I have an identical problem and don’t know what to do about it! Hair sounds the exact same as yours. Is the Olaplex permanent, will it wash out eventually? I would love to know the outcome and what worked?
I had Olaplex yesterday, as part of my conditioning blowout. The first and last time, I will go anywhere near this product. The result was terrible…. It left my hair dry, dull, frizzy and damaged looking. I’ve been applying serum, to help add back moisture .. I use Iden Bee Propolis Silkshine .. the pump not the spray.. I usually apply just a couple of drops… but the Olaplex has dried out my hair so badly…that I’ve been applying it at intervals during the day…hoping to halt further damage from the Olaplex… I just put a couple of drops in my palm rub my palms together and then run my hands over and through my hair and on the ends..
These comments blow my mind! Obviously whomever applied the treatment didn’t do it properly because of the bond chemistry. Was it put on your hair dry? Left in then step 2 put over step 1? Geez!
Hi everyone,
I would be more than happy to speak with your stylist directly and help troubleshoot as Olaplex itself is not going to dry hair out. Feel free to email me at jordan@olaplex.com
My hair has been on a colour lap of victory this last year, bleached blonde to pink, to copper to brunette and now, thanks to Olaplex back to bleach blonde. It should have fallen out, it hasn’t. It looks healthy, it shouldn’t. This is all down to Olaplex used in the bleach and then at home treatment used every week. I’m a complete convert to it.
29 yr. PHD. (Professional Hair Dresser) with strong color and texture experience. I use and endorse Olaplex whole hardily, never had such great consistent results. There is a method of use and application. But just like other services that suffer ill results at the hands of uneducated practitioners, Olaplex is not exempt. If you are taking away from the hair, you need to put back what you take. Just a small example: Your Color retouch can be performed without Olaplex, But the shaft and ends of the hair can be treated with Olaplex to repair and hold color longer. However, if you add the history of what you might do to the hair at home, previous chemicals and cheap harsh products you may subject it too. Then you would need to Olaplex 2 o 3 times a week. The #3 Olaplex treatment is for home maintenance.
So you’re all right!! it’s not a miracle product… just a miracle repairer the likes I have never seen before. 🙂 thank you for your articles!! First time I visit 🙂 glad I did. will share forward.
Bismaleimidoethoxyethane crosslinkers have been used in other industries for some time, and they do form stable thioether crosslinks that are not cleaved by reducing agents, and hair coloring is not a reducing process.
The reaction is very specific to pH 6.5 to 7.5 and 1,000 times slower at a pH above 8.0. Since oxidative haircoloring is performed at a pH well above pH 8.0, I question how bonds would form when the product is added to haircolor? The atomic weight seems to be about 246 amu. That’s big. Most haircolor ingredients are below 100 amu. I question how this can penetrate into the cortex and fix the bond?
So, as it seems it’s only coating the outside of cuticle. Basically, the whole system works just as an intense conditioner to give you less damage when coloring and nothing more.
Are the linkers in those other processes Michael acceptors, or do the thioethers form by some other mechanism? Michael additions are generally not very pH-sensitive AFAIK.
246 amu is plenty small enough to penetrate the cortex – coconut oil (~700 amu), for instance, penetrates.
I only tried Olaplex once and will never use it again. It wasn’t applied with color but as a treatment with a blowout.. It dried out my hair and left the ends frizzy and horrible looking. I applied tons of serum as soon as I got home, to add shine and moisture back to my hair. And snipped the ends off, because they looked so bad.
I was so upset that I even canceled the date, I had planned for that evening.. I can honestly say, this was the worse blowout, I’ve ever had. Dry, frizzy and damaged looking. Not the usual sleek, shiny results achieved using a regular conditioning treatment as part of my blowout…
Enough reviews have mentioned similar results as mine. To at the very least serve as a warning… If it dries your hair, like it did mine..My advise is to never use it again and risk even further damage..
I can only hope that applying a ton of serum has helped to halt the damage the Olaplex caused…
I had an Olaplex treatment done towards the end of last year (2015). My hair was medium-dark brown (salon dyed) and I wanted to lighten the ends. The hairdresser I went to suggested Olaplex as it wouldn’t damage my hair as much as bleach. (A little back story – I did some hair modeling up until 4 years ago, and my hair was always extremely healthy, even with the harshest bleach my hair always looked and felt very healthy). D-day came and I went in for the treatment, the hairdresser put the Olaplex in for what felt like a very long time. After a wash and a blow dry I saw the disaster that is now my hair. Firstly my hair is ORANGE and secondly it feels like end of a straw broom. I am absolutely heartbroken and I have warned everyone I know who was planning on going for the Olaplex treatment to stay away! I now have to cut all of my damaged hair off (after growing it out for years).
I had the exact same result. I’m heartbroken and babying my hair like crazy with masks and serums. It’s so dry, splitting and breaking off. I wish I’d never used Olaplex no 3.
Hi Petra,
Olaplex itself is not going to turn hair orange. This generally means too much product was added into the lightener which hindered the lifting process. If the ends of your hair feel like straw, this would be due to some form of overprocessing. Olaplex is an amazing product however it will not make hair invincible. Please feel free to email me and I will gladly help you and the stylist troubleshoot.
jordan@olaplex.com
I’m confused. If too much product hindered the bleaching process then how can her hair also be over processed?
I just want to say THANKS for the info. As a biologist, I think like a scientist but I don’t have a strong enough background in chem to do what you did. I have been getting keratin for at least four years now for my very frizzy gray hair and I love the results. There is no significant damage to be repaired but I was thinking that Olaplex 3 might be a way to extend the results for a few weeks before I would normally get it done again, and I’ve got some frizzy new hair to tolerate. I thought, based on the way it works, that it wouldn’t be useful at the beginning because the keratin has sealed the cuticle. But it seemed like a good option to use for 3-4 weeks at the end. Perhaps even replace the expensive keratin treatments altogether.
So now I know, and I won’t be trying this product. At age 60, I’ve lived through dozens of these frizz cures – oils, silicones, Living Proof (minimal difference), etc. Keratin is the only treatment that actually works, and at the price of losing my beautiful waves for a few weeks. But after that – well, people are constantly complimenting my hair. Total strangers. That never happened before keratin. I had major league bad hair.
And I’ll bookmark this site – which is a huge compliment to you, because I’m a total skeptic and it takes a lot to convince me that you know what you are talking about.
Thank you so much! 🙂
Hi,
I haven’t read all these comments but they are interesting and interesting post too. I want to let you know my experience. I am between virgin level 1+ 2 fine hair. I bleached it with cheap blue bleach powder and 30 vol with foils. I foolishly left on for an hour. I washed out and have left it to allow it to rest for at least a month before bleaching again to get desired pale yellow. At present its not the best colour but I can live with it as I put a toner on it after. I then came across olaplex and I DID buy from EBAY. Prior to my olaplex application I shampooed hair with clarifying shampoo, and prior to that for a few days I was fading the toner with vitamin c treatments. After clarifying I then put step 1 on hair wrapped it in clingfilm and left for 30 mins then I used step 2 immediately ontop of step 1. I wrapped again in clingfilm and a head wrap and went to sleep. Next morning I shampooed it out and did a home made protein mask and then another light clarifying shampoo and ‘cone’ free conditioner. My hair feels amazing and it is actually feeling silkier as time goes on. It is less dry and is definitely improved. I also did a strand test with olaplex added to the bleach on a strand of hair previously bleached and a strand test 2nd round of bleach on previously bleached strand without olaplex. The bleached hair strand that was bleached second time with olaplex was in much better condition than the one without the olaplex. Additionally the one with olaplex lifted better and quicker weirdly. I used Goldwell bleach second time round.
All I can say is, I bought olaplex from EBAY, and I am glad I did, the girl was a hairdresser and is selling off to make money obviously but it worked for me so I don’t care, it saved been ripped off in a salon, its easy to do at home, and I am going to use it again before I do my second bleach application to my hair. Olaplex works if used correctly.
I have been using Olaplex now for over a year. I can honestly say that there are good times to use it and not so good times to use it. I think people who are not in the hair industry nor those who are not chemists don’t understand the basics of the composition of hair and that could be why they are getting dry, frizzy, unruly hair.
Olaplex is NOT a conditioner. it is a bond reformer. It is not magic. It is science.
I don’t use it on every single color process I do, but I have used it with great results. I have seen some hair actually go back to its former crappy self after stopping use. But I have also had great results when dealing with hair that is super compromised.
Hair coloring is not like painting a wall. and those who are not professional don’t understand the science behind it. They don’t understand that the disulfided bonds only make up a small % of their actual hair strand itself. The other properties that really are being problematic here are moisture and protein. Olaplex doesn’t add either one of these things and If they are seeing dryness and breakage it is probably because they need a combination of both along with their Olaplex treatment.
I don’t use it on everyone like I said, and I believe that prople are really pushing the envelope and expecting it to do more than it actaully does.
I will continue to watch and see but I do think it is a great tool in my coloring tool box.
Do I need a protein treatment (like Kerastace Vita Cement) after highlights If I am using Olaplex system steps 1, 2 and 3?
Hi Patricia. Protein itself is an entire separate issue. If you notice the hair is gummy, this generally shows a lack of protein within the hair. If hair is dry, this shows a lack of moisture. Olaplex deals with broken disulfide bonds. All issues within the hair must be addressed in conjunction with one another.
jordan@olaplex.com
I don’t mean to be rude but are people not at least reading info from the Olaplex website because there are tons of people that think Olaplex does what protein & conditioning treatments do. It’d be great if your comment popped up everywhere Olaplex & moisture/ protein are mentioned!
Hi I had a simular bad experience with Olaplex using only step 3 and as stated you should be able to use alone. My hair is blonde on top and light brown (color treated)on the bottom. My hair was in fairly good condition.
Both times my hair felt good right after treatment then when I shampooed it it became dry, brittle and hard to manage. I now have breakage in one area( in the back interestedly not where it is blonde) and have finally gotten my hair back to its original state after intensely conditioning for months. I never had this problem prior to using Olaplex.
I noticed the only answers to the many people who have had the same result is that they did not use color/bleach correctly or used Olapex incorrectly and no one has addressed why so many who have used it had this problem, as it is not just incorrect coloring/bleaching if the product was the ONLY change in procedure. Would like to hear from a representative of theirs a better answer ( not just BS) as to why so many have had this happen.
So you actually say that you did it yourself, and only used step 3 – could it be the issue from bleaching it in the first place??
It sounds like you didn’t actually have it done in a salon from what you said, re: only using step 3 (which could explain the ‘so many problems’). It also seemed like a lot of the previous issues were when people were using Wen, which has it’s own massive issues.
Personally, Olaplex has been a godsend for me; I don’t bleach my hair, but I do color it– the problem I have is that my hair is curly and fine, and it likes to tie itself into knots. When I use Olaplex, it no longer mats itself together on the back of my head, and the ends don’t tie themselves in knots anymore. I got my last color/treatment about 3 months ago (march 10), and I’m getting another one on Wednesday, because it’s now started knotting back up again. It just makes me less frizzy/knotty.
I don’t think I would use the take-home treatment, since just the one seems to work for me – but maybe it’s that the take home treatment thing isn’t strong enough? =/ idk. I wish you best of luck, though!
Hi Michelle. Feel free to email me directly and I will help you troubleshoot.
jordan@olaplex.com
I used Olaplex on my hair and it has been fantastic. My hair is softer and stronger. For those who say the texture changes, yes, I did notice that a bit. My hair feels thicker (and looks thicker and more dense). It did feel a bit dry which I believe is from BLEACHING. However, I used a good bleach and my hair didn’t feel nearly as dry as it should have after doing a second bleach process. I have not had stretchy or mushy hair when it is wet or breakage when it is dry. I did a lot of reading beforehand and I followed the Olaplex instructions to a t.
I believe that Olaplex saved me a TON of damage and I would never do what I did without it. I also know from reading and research that I cannot consider Olaplex to be a conditioner.
Because it is a little dry due to the BLEACH I used, I am doing deep conditioning treatments, and I am using moisturizing and protecting products as anyone would (should) if they bleached their hair. I also wash it only once or twice a week and I let it air dry; on occasion I flat iron it but not without products for heat styling. Olaplex doesn’t mean you don’t need to take care of your hair…or that it doesn’t need moisture (I live in a desert climate, so I usually need it anyway). Olaplex it is NOT A CONDITIONER.
Olaplex likely did not cause your hair to snap off and as someone said, it’s probably actually why you had hair left. It doesn’t mean you can just abuse your hair without any regard to limits. You still have to watch your hair carefully and there is a LIMIT to what you can do!!
But compared to past double bleach processing, my hair is in much much better condition. It feels like I didn’t really do anything to it. I also have permanent extensions in my hair – just one row – but they even feel silky and stronger than before.
If Olaplex did nothing, do you really think people would rave about it so much?? Just look up videos on YouTube and you’ll find many more successes with Olaplex than failures. You’ll also see and read that many stylists will not do bleach or color treatments without it.
If one more person says that Coconut Oil can do what Olaplex can do, I might flip out. These are usually people who have never tried Olaplex who are quick to judge. I have tried both and I love coconut oil for both my body and my hair – but coconut oil is not Olaplex. They are both helpful but they do two very different things.
I just wanted to make a statement FOR Olaplex after reading so much anti-Olaplex crap. For me it has been fantastic. After two months my hair has not fallen out so I’m going to venture a guess and say that I think maybe it’s going to stay on my head.
I have dyed and bleached my hair to a pulp over the years and I am trying to regain my natural healthy hair. As it has taken years of abuse I realise this will not happen over night. The Pureology Conditioner does work a bit, but the Somaluxe Argan Oil is what made my hair looks like I am 20 years old again! I massaged it into my scalp and hair and have left overnight and washed out in the morning, my pillow wasn’t stained or greasy looking so thats a relief.
You can feel the difference, my hair doesn’t feel as coarse and dried out. I blowdried it and it felt great, shiny and looked healthy and sleek. I do recommend the Somaluxe Argan Oil as a deep conditioning treatment once or twice a week to anyone who has dyed hair. The bottle isn’t very big but you don’t really need a lot, I would estimate this to last me atleast 3 months
Any one compared olaplex to the uberliss bond treatment ?
Since I’ve had an olaplex treatment in a salon my hair now feels like it has a residue or a coating on it (like some other commenters said) does anyone know how to reverse the olaplex treatment? Or how to make the olaplex coating feeling go away? Thanks, Ro
Hi Rose. If your hair feels like it has a residue on it, this is generally due to some type of buildup. Was the hair clarified or shampooed prior to application of Olaplex treatment? This is extemely important as oils, silicones and other film formers will act as a barrier. I’d be happy to help you personally.
jordan@olaplex.com
Hi, Jordan.
Can you recommend specific products that clarify hair properly in preparation for Olaplex? Since there are various methods (shampoos, treatments) & since Emily’s comments have been echoed by others I want to feel confident that I’m using an effective product that works for the Olaplex.
Additionally, I think your clarification suggestion & the fact that you don’t recommend us a cleansing conditioner like Wen with Olaplex should be added to the Olaplex facts page. I don’t recall seeing info on either issue except for here in this article. I’m glad I found this article & am thankful for Lab Muffin and the commenters because I would’ve used my Wen but not clarified.
Thanks for your help.
Jordan,
I was just made aware that the original Olaplex FAQ page is gone & has been replaced with a help page. While there is a “cleansing cream” section it doesn’t mention that we shouldn’t use it to wash out Olaplex. It states to clarify prior to using Olaplex if we’ve washed with a cleansing cream. However, I think there should be an additional “prep” section that recommends clarifying as well. Otherwise, people will only know if they’ve read these comments or clicked on the site’s cleansing cream section.
For everyone posting questions for info please look here & do so REGULARLY! Had I not done so today I wouldn’t have known that NOW Olaplex says to NOT BUMP UP DEVELOPER WHEN BLEACHING & to only use 20% ON SCALP like most bleaching products have suggested.
http://help.olaplex.com/hc/en-us
Thank you for the update that Olaplex changed, well pretty much all the basics. The upping of the developer and it seems they also changed the amount of Olaplex 1 you should add based on the amount of bleaching powder. It was 15g = 3,75 ml and 30-60 g = 7,5 ml but now it says 30-60 g = 3,75 ml. Have they changed their formula?
I’ve recently received an email from Olaplex saying:
“OLAPLEX no longer requires you to bump up developer or increase processing time.
OLAPLEX from the very beginning, have always respected your feedback and with your input we have decided to make adjustments to our directions of use based on your real world results.
What does this change mean for you?
OLAPLEX no longer requires you to bump up developer or increase processing time. We want you to use half the previous amount of Olaplex No.1 while getting the same amazing results and get twice as many applications. This cuts your cost per application in half. In most cases, stylists are able to go well beyond the 140 applications.”
Hi everyone!
If anyone is having issues or has Olaplex related questions, we would be happy to address them directly. Olaplex has now been used more than 60 million times in over 300,000 salons worldwide. Our goal is to help everyone achieve beautiful, healthy hair.
Jordan Alexander
Vice President of Education
Olaplex LLC
jordan@olaplex.com
So I spoke with Olaplex about your misinterpretation article on Olaplex and Dean states this:
Directly from Dean regarding this article: I wrote to lab muffin. She was partially correct but not quite understanding the mechanism of ionic bonding vs. covalent bonding. Yes, covalent is stronger but when it breaks which it easily does in the hair, it’s broken forever. Ionic bonding is still strong but relinks to other ionic bonds when they break constantly reforming new links. This is part of our technology that is permanent. Lab muffin is a skilled chemist, but understand that we have thrown many methods of interactions of our chemistry and we are not trying to make it easy for an ordinary chemist to comprehend and attempt to copy or work around. If you fail to use olaplex, you will interact sulfur hydrogen bonds to oxygen creating more sulfate groups that damage your hair. Olaplex prevents this action a vast majority of the time. If Olaplex were not permanent, hair would be breaking off in clumps after a few shampoos like imitator brands. It doesn’t because of ionic bonding. This is what lab muffin failed to understand or forgot to write. Please ask her. She should be able to clarify. Great question. Dean.
Please if your gonna write a article that might show faulty info then find out what you need to know explaining said product please retract the FALSE info. As many readers stated they had bad experiences with Olaplex, mass majority are due to lack of education on product and stylists mishandling product and not following directions or using their degree to its potential then blaming the product. I find your article and comments disturbing, as I do not see a rebuttal or update on it after you spoke with Dean clarifying to your readers how it works. Olaplex HAS AND IS changing our industry. I say grasp it and let’s see what other amazing things we can do!
Dean never wrote to me, I actually have zero results in my email inbox for anyone named Dean emailing me. If you read his comment carefully, the only bit where he disagrees are the last two sections of “Things That Are Bugging Me”, which is speculative and has zero impact on how Olaplex is used or how it works. Please reread his comment and this post before being “disturbed”. Thanks!
Wow, it looks amazing! Now I wonder how protein conditioners and keratin treatments works and how they compare?
I know that with those you can have too much, making your hair brittle and dry. Is this treatment suitable in a situation like that? Your friend in the picture has dry hair in the before picture but supermooth hair in the after picture, it even looks moisturized..
I found some info on it. The protein treatments only work on the cuticles. Does this product perhaps work on a deeper level or is the main difference in the mechanism? And is it a common effect that it revives curls?
My long, frizzy, curly, damaged, bleached to death dry hair has never felt better than after using Olaplex! Olaplex no. 3 left in the night before I wash also makes a huge difference. I want to buy it in bulk, it is excellent!
Did you know that Schwarzkopf claims that their Fibreplex does the same as Olaplex? But after questioning I found out that their active ingredient is magnesium citrate. Nowhere can I found support for the benefits of topical application of magnesium citrate on hair, let alone that it could repair sulfide bonds. Seems like a scam!
I have a question about bimonthly use of Olaplex without any hair coloring/perm/chemical services of any kind. I am 55 with fairly healthy full set of salt and pepper hair. I use Kerastase shampoo daily and Pantene/Jhirmack purple shampoo for silver hair bimonthly. I also use Kerastase conditioner everyday (i use very small amount and leave in hair everyday).
Tell me, am I overkilling it or am I insuring myself that I retain my full set of hair 10+ year from now.
@Trina B – pls contact me via e-mail if you managed to do something about it because I experienced exactly the same problems like you. This is really a nightmare and I’d like to sue them as well. My hair was thick, healthy, shiny, natural…and now, after using Olaplex1,2 and 3 there is no volume, it’s stripped with orange, fuzzy at ends, oily, thinner and thinner every day…I can’t stop crying, looking at the mirror and touching straw on my head:-(((! Thanks a lot
thank you for sharing this hair treatment. i really need this thanks!
I have used this product once. I left it on for about half an hour to give it a chance to work. And it really has. My hair feels quite silky and smooth for a change.
What do you think of Wellaplex? It’s supposedly active ingredient is malic acid, which I highly doubt would work.
I agree!
Olaplex has literally been my worst nightmare for my hair. It turned my soft, shiny, smooth hair into rough tangled crinkly strands with a SINGLE STANDALONE TREATMENT. NO BLEACH OR COLOR. I left it on for 4 hours and I remember the panic as I rinsed it out my hair felt like rough (like holy crap ROUGH) and so knotted together and it completely stripped all shine and it feels like its coated in something and had lost all volume. Its been over 6 months and I still want to cry because my damaged hair is breaking more and more.
The product should seriously have more warnings on it. This Jordan guy is totally delusional or ignorant to think it can’t damage hair when so many people have complained about the same disaster I have experienced. I struggle to style, brush, manipulate my hair in any way. I want my pre-olaplex hair back sooo bad. Patience girls, just gotta let it grow 🙂
To get the best outcomes possible, it’s vital to utilize Olaplex when applying a colour treatment, as opposed to doing it afterwards. People often see Olaplex as a miraculous solution for any type of damaged hair and assume it would be fine. In any case, it is an experimentally created treatment that does its work from the inside, rather than a conditioner that enhances just the shallow state of harmed hair.
Hello,
I don’t know where else to ask this, I’d be very happy to have an answer.
My question is : Is olaplex a convenient, colorless equivalent to henna? Apparently henna also uses michael reactions to bond with the hair.
Would you have an opinion on that?
Thanks!
I’m surprised of the several negative responses to using Olaplex. There really must be quite a bit difference of results based on hair types or hair damage, or maybe there was lightening or bleaching that was bumped up too much because Olaplex was getting added. I had dry, brittle damaged hair from over 30 years of coloring and bleaching and Olaplex was amazing! I love it and it was a miracle on my hair! It’s so soft and silky. It feels the best than it has for many years! Before I always had to put other products in my hair to keep it from looking frizzy. I used the full 3 steps, but didn’t color or bleach my hair with the first step as the directions state that you can do. For me it really was like magic. <3
Can you compare Olaplex No.1 to Ion Absolute Perfection Booster Step 1 and Continuum Ro-Maxx? I have heard the Continuum is a similar bond rebuilder like Olaplex, but not sure if Ion truly is. Some say it is more like Redken pH Bonder, where it just helps seal the outer layer of the cuticle, making it feel smoother and softer.
Ion Absolute Perfection Booster is being called a dupe for Olaplex. Looking at the ingredients, it does not look like there is an active ingredient that is proven to link disulfide bonds like Olaplex. I am assuming this means the Ion does not work like Olaplex, but is more of a protein treatment that helps seal and fill in the outer cuticle like Redkin pH bonder, B3, etc. What do you think?
Now I know how Olaplex works and how perming works, I wonder what the effect is of the way you keep your hair while it gets absorbed. By that I mean straight (like most people will) or with some waves squeezed into them. Because I have wavy hair and when applying to combed and thus straight hair, I didn’t notice an improvement in curl texture, maybe it even curls less now. Next time I’m going to try keeping it wavy during the treatment time!
Perfect article! Thanks for sharing. It is really great to hear from an expert and see what really goes on, on a molecular level.
Just curious if the last step can be done at home without the first two steps, what would the results be. I haven’t tried Olaplex yet but have heard a bit about it recently and got curious. Thanks for the explanations.
That’s usually what’s recommended – I personally think you may as well buy Olaplex 2 rather than 3 if you can find it, since it’s usually cheaper and has more of the active ingredient.
Hi, Michelle:
I’m a deliriously happy Olaplex user. I do it myself. I have fine, kinda thin, kinda old, color-treated wavy hair, and I wash my hair with detergent shampoo before doing an Olaplex treatment as instructed. Then I leave the Olaplex on overnight (unless I added it to my dye!), and shampoo it out, again with a detergent-based shampoo, then next morning.
The rest of the time, I wash my hair every two weeks with conditioner. (By the end of two weeks, it begins to get a little oily at the roots, but that’s the extent of it.)
Question: I notice from the ingredient panels you listed (thanks ever so much for that!) that the conditioner contains some Olaplex in it. All the directions for Olaplex say you must wash your hair before using Olaplex and afterwards, to get it out of your hair. If it’s in the conditioner, though, it’s not being washed out, obviously. This isn’t a problem?
Another question: As someone who doesn’t want her hair to “squeak” or be a tangled mess after shampooing, is the Olaplex shampoo, which appears not to have a lot of detergent in it, gentle enough for someone who washes her hair with conditioner? Can you tell from the ingredients list? If my hair isn’t gonna feel soft and silky after using it, I’ll give it a pass.
To the folks with the problem hair after use: the instrux say to use a protein treatment after an Olaplex treatment. Not sure it’ll help (some of the folks here seem to be in dire straits, and I was sad reading about their results), but figure it’s worth mentioning. My formerly dry, broken, frizzy-ish hair is shiny, silky, and young-looking again, so I’m crazy about Olaplex!
I think it’s fine for Olaplex to stay in the hair – I think the washing out is just because some people think it’s a conditioner and complain when it doesn’t leave their hair smooth.
Also I think if your hair is good with co-washing, I’d stick with that. Pretty much any shampoo is going to be harsher than a conditioner, and you’ll get enough Olaplex from the other products anyway!
Also thanks for the broken link warning 🙂
Last question for today, Michelle. I swear!
It’d help all us hair freaks if you’d contrast and compare INNOluxe ReBond V2, who was sued by Olaplex (and lost) with Olaplex. They claim to not only repair hair on the inside, but also to make it shiny on the outside. I sure would love to hear whether there’s any validity to their chemistry. I’m thinking there must be, otherwise there likely wouldn’t have been a lawsuit.
I already use Olaplex 2 at home instead of Olaplex 3 and have seen great results. I’m considering doing the regular stand-alone treatment (Olaplex no. 1+ Olaplex no. 2) but I am not sure if the results will be THAT much better than just doing Olaplex no. 2. Do you have any information or advice on the expected incremental benefit of adding No. 1 to an existing No.2 routine?
Thanks,
Bora
I have hair similar to your grind Mary’s “before” photo (but not quite as undefined). What at home consumer products could I use to revive my curls? Should the Oliplex (or otherwise) product be used long term or only occasionally? Thx!
This is an awesome blog! Thank you for informing the public with genuine scientific insight. I studied biological anthropology in my undergraduate and always want to fact check products through a scientific lens, but the scope of my education isn’t broad enough to be able to get into the nitty-gritty chemistry of everything. This blog is like having a best friend at a university who also cares deeply about beauty products (something I’ve been looking for my whole life, lol!) and can explain everything to me in terms that I can understand without having to hit the books myself. Thank you! Thank you! Thank you! You should write books about beauty products. Please keep doing what you’re doing.
This is where some of the confusion around the hair treatment comes in – the Olaplex hair treatment is not a cure-all for dry or damaged hair. It is a scientifically-developed treatment that works on hair condition from within, as opposed to a conditioner that improves only the superficial condition of damaged tresses. Olaplex is also split into three main stages, and when you use each one differs, with four hair maintenance products to support your hair condition.
I am ver dissapointed that olaplex claim these products have no toxins, hewever the first one i look up PHENOXYETHANOL is toxic to kidneys, liver and long term use can lead to organ failure!!!!
Great info, thanks!
Isn’t Olaplex mostly just Plastic? I don’t understand why no one says it straight. It is “rebounding” the hair with plastic. If you remove the plastic you are left with very damaged hair. It is not a true rebounder. I just don’t feel right about putting plastic on my clients hair and scalp
Bis-aminopropyl diglycol dimaleate is not plastic. Silicones can be polymers, but protein is also a polymer…
I have Asian bleached hair (level 7).
I was attracted by the marketing of Olaplex. Unfortunately, after 6 months of use, I was disappointed. The Olaplex no. 6 forms a silicon layer like a wax coat that made my hair very dry and dull.
I recovered my silky hair only after a couple of clarifying shampoos and went back to my Kiehl’s Amino shampoo (paraben-, silicon, sulfate- free) to keep my silky hair without conditionner.
It is worth reviewing this No.6, it has a lot of dimethicone …
Hi Michelle,
Do you have any info on how the K18 hair treatment works? Olaplex promises to repair the disulfide bonds that have been damaged, K18 seems more protein-based. Is it something comparable or just marketing?
Thanks!
It works in a different way but it’s really promising! I might do something on them soon 🙂
Yes! Looking forward to a K18 video!
This is awesome!!
You’re too smart!!!!
Can you please do one for ColorWow?!?
I wish I never used Olaplex. I lost 1/2 my hair and it made the leftover 1/2 so brittle, dry, crunchy with a waxy feeling on the hair and scalp. Beyond horrible. Please do your research before using it and see if anyone else has had the horrible results that I have. I’m crushed.
Olaplex has been amazing on my virgin hair having spread out the 2 step system that I got off ebay way back in the day over years. Some of these comments simply seem stupid and don’t understand that hair starts to accumulate damage the second it leaves your scalp and is exposed to the world. If you’re bleaching it, it’s still going to be hyper damaging, just maybe less hyper damaging. And this is why the brand has to protect itself from you people and leave poeple like me who would never be caught dead in a hair salon having to buy it from 3rd parties. Thankfully, that’s actually no longer the case as legitimate US retailers now have it online.
As with other famous topical dermal ingredient, the red tape means it’s legit.
Also saves you a lot of money you’d otherwise spend on gimmicks when the truth is you just have long, fragile hair.