I’ve recently started trying out thermal cleansers. These are special cleansers that heat up when you rub them on your skin and add water. What’s the point of them and how do they work? I’m glad you asked, hypothetical reader!
Do pores open with heat?
Heat makes muscles relax, while cold makes muscles contract (you may know this from treating sore muscles from exercise with wheat bags, or just from soaking in a hot tub!) However, pores don’t have muscle, so heat doesn’t open pores, and cold doesn’t close pores.
Then what’s the point of a thermal cleanser?
Even though heat doesn’t open pores, it can still help you clean your pores!
1. Many fats and oils turn from liquid to solid (and back the other way) at around skin temperature. For example, shea butter melts at around 40 ˚C and coconut oil melts at around 25 ˚C. That little bit of heat could help dislodge some solid lumps of too!
2. Liquids also get thinner with heat (the scientific term for this is ‘less viscous’ – it flows more easily, like warm honey, or oil in a hot saucepan). Adding extra heat would make the oily gunk in your pores thinner and more easily able to flow out.
Of course, there are lots of other ways to add a bit of heat to your routine – warm water, warm compresses and a bit of light steaming will help too! However, excess heat can cause burst capillaries (ugly redness in your skin), so it’s best to be cautious.
Thermal cleansers are also pretty fun to use! I’m a sucker for a sciencey innovation, and it’s pretty cool feeling the cleanser warm on the skin.
How do thermal cleansers work?
Thermal cleansers typically contain ingredients which heat up when they dissolve in water. The typical way you can measure how much heat is coming out is by seeing how much it heats up a known quantity of water, in a very sciencey coffee cup:
No, seriously, chemists actually use coffee cups to measure this sort of thing!
Here’s what you do:
1. Put a set amount of water in the coffee cup. I used 20 mL, measured by syringe.
2. Measure the temperature of the water.
3. Squirt in a set amount of the thing being dissolved (in this case, I added 1 mL of thermal cleanser, also measured by syringe).
Here’s 1 mL of Nivea Pure Effect Thermo Daily Warming Cleanser:
4. After squirting in, you have to stir it until the whole mixture is mixed thoroughly (no lumps sitting in the bottom!)
5. Then quickly measure the new temperature. For Nivea Pure Effect Thermo Daily Warming Cleanser, 1 mL of cleanser heated the water up by an average of 1.6 degrees (I ran each experiment 3 times, and the results were within 0.2 degrees).
For Dermalogica Multivitamin Thermafoliant, the water heated up by an average of 1.0 degrees (averaged over 3 runs, results were again within 0.2 degrees).
If you’re into this sort of thing, you can actually calculate something called the enthalpy of solution, which lets you compare the heating effect with other chemicals. I refrained before my bathroom exploded with excess nerdery, but if you’re super curious (or if you’re studying for a thermochemistry exam), here’s some info.
So now that we’ve established that they both do heat up (i.e. the warm sensation is actually warmth and not chemical burning, for instance), and are activated by water, and that the Nivea one is a bit more warming than the Dermalogica one, how about on the skin?
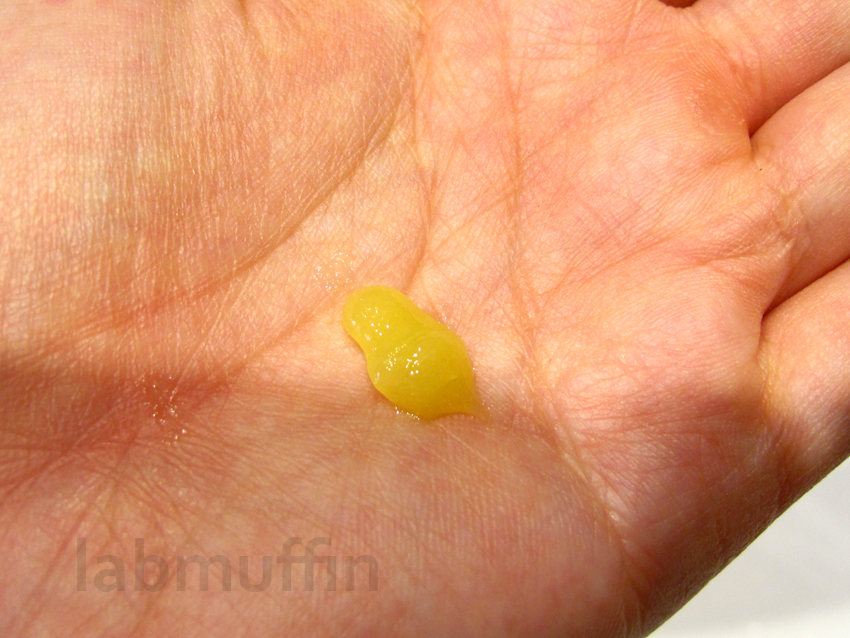
Realistically, I’d only apply about 0.2 mL on my skin in one go – about this much:
Now, here’s where the experiment started to go south. Unfortunately, the warm sensation only lasts for a couple of seconds, which isn’t long enough for the mechanism inside a regular thermometer to expand and register the temperature change. By the time the thermometer reached the top temperature recorded, my skin was feeling clammy. I would estimate that the maximum change wasn’t more than a few degrees though.
This experiment would probably work a bit better if I had, say, an accurate infrared thermometer, which wouldn’t rely on slow expansion. I can dream!
What are the ingredients responsible?
So what’s the secret ingredient that heats up? There are a few ingredients that are used for the warming effect – in Nivea Pure Effect Thermo Daily Warming Cleanser it’s PEG-8, glycerin and PEG-150 (the first 3 ingredients), while in Dermalogica Multivitamin Thermafoliant it’s PEG-6 and magnesium oxide.
Unfortunately, the magnesium oxide (as well as sodium bicarbonate) in the Thermafoliant pushes the pH of the product to a skin-unfriendly pH 9. Since it’s a scrub, it doesn’t stay on the skin that long, but I’d be cautious about using it on sensitive skin or more than once weekly.
The Nivea thermal cleanser doesn’t rely on alkaline salts for the heating effect, and is at a much more skin-friendly pH 4-5. I particularly like it since it’s a gel type cleanser, and glycerin always makes my skin feel ridiculously plump and hydrated. It also contains magnolia extract which has shown some promise as an antibacterial in cell experiments (though not on skin), and offers some UVB protection as well (that’s the sunburn and cancer-causing type of UV radiation). The marketing relies a bit on the terms “pure” and “impure” to describe skin which irks me, but that’s a rant for another day!
These products were provided for editorial consideration, which did not affect my opinion. For more information, see Disclosure Policy.












Love the science!! Thank you 🙂
Yay! *throws the science*
Very interesting post haha! I actually learned about enthalpy last term in chem 🙂 I really enjoyed your little experiment, loved the nerdery! I’ve always been skeptical about people who say to ‘open and close the pores’, I’m glad you’ve cleared that up for me! I’ve never tried a thermo cleanser before and they seem intriguing but I think I’m okay with just using warm water for now, haha!
Tasha // shiwashiful.
I think thermochemistry was my worst subject – anything physics does my head in!
Enjoyable review… Experiments took me back to my lab days 🙂
Thanks! Though to be honest I don’t think I’d stick something from my lab on my face, ever… I don’t trust what other people are putting into my air 😛
Amazing post! Cheers and have a great weekend 🙂
Thank you 🙂
Very cool! I knew pores didn’t really open or close with temp (otherwise I’d look FANTASTIC after spending an hour outside in the winters around here..!) but I had my “ah-ha” moment when you mentioned heat making oils flow more easily – duh! I don’t know why I hadn’t thought of that before. ;P
I’ve been thinking about oils a lot lately, thanks to oil cleansing – a quick rub in a warm shower and the oil comes down to a normal level when I pat my face dry with a towel. Insanity!
I love when you talk science – it actually makes sense, unlike my teachers back at school! Thanks for a really interesting post 🙂
Amy | Bubbles & Blush
My theory is that no one actually hates science – they just haven’t had the right teachers 😉
Oh hey, I bought a thermal mask a while back and I was wondering if you’d review something like that! I find it quite lovely but I wasn’t sure if it provided any real discernible benifits.
I think it really depends on what you’re doing to your skin the rest of the time – if you’re using it when you’d otherwise be using cold water with a regular cleanser, then it could definitely help. If it’s a well formulated mask otherwise, it’s probably be worth using anyway!
So interesting. Thank you!!
I’ve been using one recently, and it’s up for review, so this is SUPER helpful to me haha!
I’m happy to help! Looking forward to seeing what you thought 🙂
Oh my god, I just discovered your blog and I’m so excited! I’ve been looking for someone who does this sort of thing!! I’m a veterinary med student myself, hoping to combine my love of beauty with animal science. Good job, your blog is awesome!
woodyxx.blogspot.com
Thanks! I bet the animal science side of beauty is pretty interesting too – all the animal products (lanolin, snail cream) and animal testing issues 🙂
so interesting