After Part 1, where I stained my nails (For Science!) to work out how best to prevent staining, the obvious next step was to figure out stain removal! I worked out in Part 1 that the teal colour is CI 77288, which is chromium(III) oxide. Here are my nails again – argh! I had to live with this for two weeks. TWO WEEKS.
The contenders
I fielded a lot of suggestions, and gathered the most popular for the experiment:
Acids: Lemon juice – This contains citric acid, and has a pH of 2-3 (quite acidic). Bases: Baking soda – Aka sodium bicarbonate (or sodium hydrogencarbonate, if you want to be super correct), this forms a moderate alkaline/base with a pH of 9-10 when you mix it fresh with a bit of water. Cuticle remover – cuticle removers are also alkaline/basic, and usually contain sodium or potassium hydroxide, which are strong bases. I’m using Blue Cross Cuticle Remover, which has potassium hydroxide (“caustic potash”), pH 13-14 – very alkaline! The theory behind using acids and bases/alkalis is that they can help some chemicals wash out, or react with colourful molecules to make less colourful molecules (hopefully).
Oxidants: Hydrogen peroxide hair bleach – This is an oxidant, a type of chemical which causes an oxidation reaction (it’s complex, but basically it’s related to adding oxygen to things). These work best on big organic (carbon-based) molecules to remove colour (e.g. clothing dye), but sometimes they’ll change ionic compounds too, like the chromium(III) oxide that was on my nails. Denture tablets – These contain a mixture of potassium persulfate and sodium perborate, which are also oxidants. They work more slowly than hydrogen peroxide.
**Safety note**
I was concerned when I found out that chromium(III) oxide was likely the stainer because an oxidised version of chromium, chromium(VI) (aka hexavalent chromium), is a carcinogen. After checking through the scientific literature, assisted by some chemist friends who handle chromium more than I do (including fellow Aussie nail blogger/scientist Shanee of Duck and Polish), I found that:
- Chromium(VI) is a known carcinogen by inhalation and ingestion (eating), after repeated exposure over a long period (think Erin Brokovich).
- It can irritate skin if it touches it, but doesn’t absorb much – there are a few reports of skin contact with chromium(VI) causing serious problems, but those involved large areas of the body.
- Hydrogen peroxide turns chromium(III) into chromium(VI) in basic (high pH) conditions.
- Persulfate is a stronger oxidant than peroxide, but acts slower (denture tablets work overnight).
- There are many antioxidants in the body which convert chromium(VI) back to chromium(III).
Given this info, I decided that oxidising the tiny amount of chromium on two nails on a single occasion was very unlikely to kill or harm me, even if my skin magically absorbed it all. I still took precautions – I didn’t let anything dry out (wet chromium won’t be inhaled but dry chromium dust could), washed my hands madly afterwards, and vowed that this would be a one-time deal! As far as I know, none of the other common nail stainers have issues with oxidants.There were a few other interesting suggestions that I didn’t have time to try, such as activated charcoal. OK, onto the experiments!
Method
To minimise skin contact with the meaner ingredients (e.g. cuticle remover), I cut pieces of cotton pads that didn’t quite cover my nails:
I dipped these into the appropriate chemical using tweezers (here’s my baking soda slurry):
Then I placed each piece on a nail and pressed gently with tweezers for better contact with the nail surface.
Here are all my fingers! L = lemon juice, BC = bicarbonate (baking soda), CR = cuticle remover, HP = hydrogen peroxide, D = denture tablet, T = whitening toothpaste (I added this at the end out of curiosity, even though I had a strong suspicion it would be similar to hydrogen peroxide since the active ingredient is usually also a peroxide).
I took off the pads after 20 minutes and washed my hands thoroughly, then took photos of the cotton pads and my nails. At no point did I feel any tingling or pain – if I did, I would’ve washed it off quick!
Results!
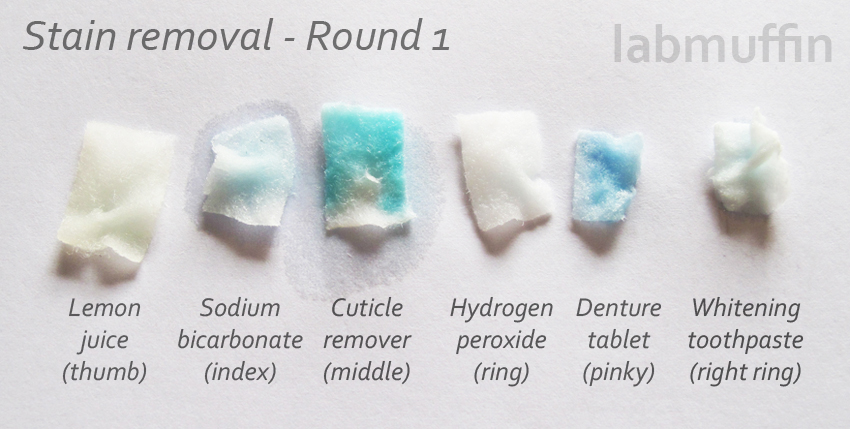
Here are the pads (the denture tablets dissolve to make a blue solution):
And here are my nails:
Here’s the before pic for reference:
And we have our winner – cuticle remover! The teal on the other nails almost didn’t change at all. From the photo of the cotton pads, it seems like a little bit of teal dissolved in the baking soda and the toothpaste which makes sense – both of these are also quite alkaline, though not as strong as cuticle remover. The cuticle remover-treated nail was also the softest, plumpest and most fragile-feeling of the nails, although it went back to normal after half an hour. It also smelt a bit like burning hair.
Why did cuticle remover work so well?
My guess: hair and nails are made of keratin. Early hair relaxers used sodium hydroxide, which is very similar to the potassium hydroxide in cuticle remover. The high pH would make the keratin in hair swell. I’m guessing that the potassium hydroxide similarly made the nail swell and become more porous (easier to penetrate), which meant the teal pigment escaped more easily. The next thing I did was try removing the stains on all the nails of my left hand using cuticle remover (including a little patch left over on the middle finger). I only soaked for 10 minutes this time. Results:
As expected, almost all the teal was gone! The cotton pads were stained teal… except for the hydrogen peroxide one, which was pale yellow. Where did the teal go?? Well, remember the ominous safety note? Some of the hydrogen peroxide must have soaked into the nail… then when I added high pH cuticle remover, the peroxide oxidised the bright green chromium(III) to pale yellow chromium(VI). Eep! You can bet I scrubbed my hands violently! Don’t do this at home, kids. It’s possible that a similar thing might happen with the denture tablets and whitening toothpaste – lucky neither of these worked that well anyway!
What about buffing?
Buffing’s always treated as the last resort – this is physically removing the top layer of your nail with a fine-grit file, and as you might expect, this weakens the nail. I used a buffing block to remove the top layer from the thumb and forefinger of my right hand:
Whoa! Almost perfect removal! It turns out that the teal only stained the very top layer – this may be different for different people though, since my nails are quite hard and resilient. I buffed the rest of my fingers, and buffed a bit of the residue off my left hand:
PHEW!!!!! Normal nails again! Success!
The best teal stain removal method
So, what’s the best way to remove teal from your nails? Well, I’d recommend…
1. Start with gentle alkalines like baking soda and toothpaste. These didn’t work that well for me, but remember, my stains were two weeks old, and I was trying for maximum stainage! If these don’t do it, try either…
2a. Cuticle remover. This is hardcore and can soften and damage your nail, at least temporarily. Try to soak it for as little time as possible to reduce the chances of that – you can try soaking a cotton bud in remover and rubbing it on your nail instead of letting it soak like I did. If you feel any pain, stop!
2b. Alternatively, if your nails are thick enough, try gently buffing out the colour.
How far the colour has penetrated might depend on what your nails were like to begin with. Most importantly, if you have blue/green stains, avoid using hydrogen peroxide unless you’re sure the pigment isn’t chromium-based!! (Other stainers are fine to remove with hydrogen peroxide as far as I know.) It’s bad for you, and it doesn’t work that well on its own anyway. If you’re extra health-conscious, avoid other oxidants like denture tablets and whitening toothpaste for chromium stains too.
I hope you enjoyed this study! I’m considering doing the same thing with other stainer pigments, but first I need to get over the psychological trauma of thinking my nails were going to be teal for a year. I have never been so grateful to have naked nails! In the meantime, feel free to throw suggestions for stainy nail polishes I should try 🙂







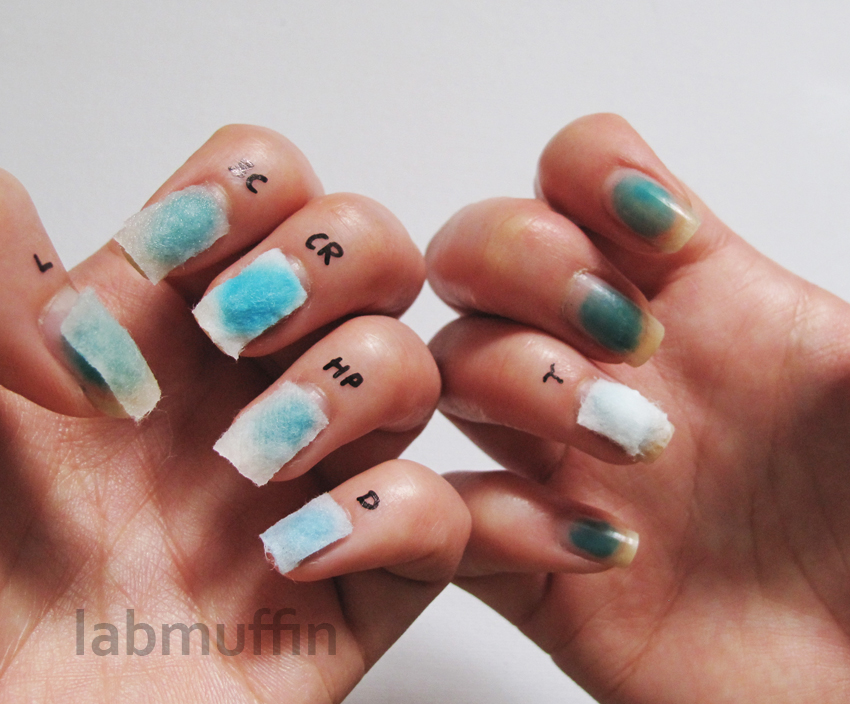



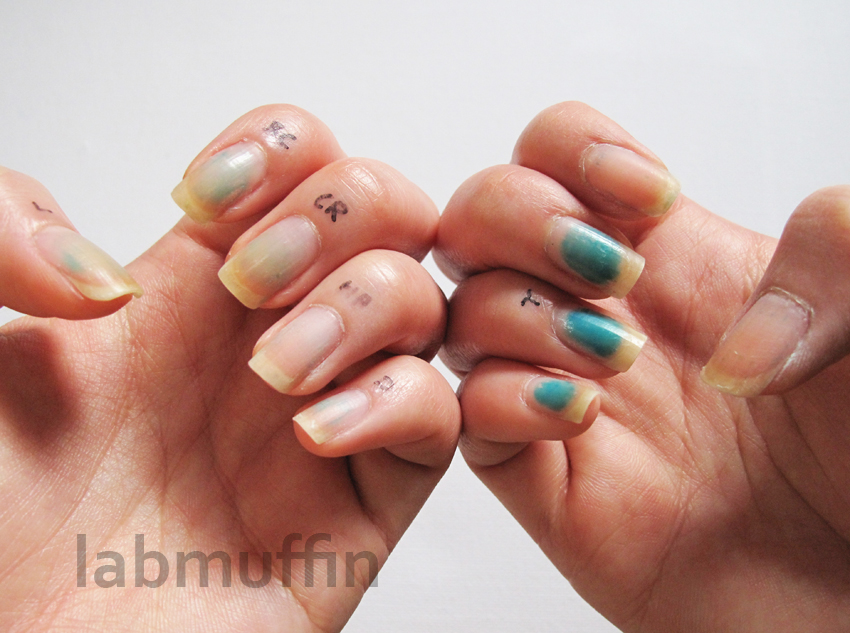








i’m truly fascinated at the lengths you go to for science – fantastic info!
Hahah thank you! 🙂
Oh my goodness! I am so glad you got all the of teal out, it would of been horrified!!
Me too, it felt like Christmas when I saw my first unstained nail! My backup would’ve been to get opaque white or nude gel polish, but I never have the patience to maintain gel manis.
I love that you were so thorough with this post! This is going to help me so much!!
Yay! I’m glad I could augment your teal-wearing experience 🙂
This is awesome Michelle! Luckily I haven’t had issues with staining yet 😀
Lucky! This is the first bad case of staining where it actually made me feel like I needed to do something about it – usually it’s just ye olde yellow ombre effect.
Can l ask where did you get the Blue Cross cuticle remover?
I bought mine from Pronail in Bankstown, but I think you can get it online (eBay etc) too.
I probably would’ve cried if I saw the yellow cotton pad :'( Putting your health at risk for beauty science – a true beauty blogger!!
(Also lol, I have the same blue and white bowls :D)
I figured it couldn’t be too bad, considering that they let undergrads use it (but not weigh it out because they spill everything = inhalation risk), and that a lot of nail addicts would’ve accidentally oxidised it before me, coupled with the fact that most of the literature I read explicitly mentioned that transdermal exposure posed minimal risks. I must’ve read over 50 toxicological reports and papers before even going near the peroxide though!
Shhh! You’re meant to ignore the technicalities and agree that you’re a hardcore beauty blogger!
Yes! Totally! 😀
Outstanding work Michelle, it’s really incredible how well you got those teal stains out. Now if you could work out an awesome removal solution for yellow staining I’ll be all set!
I’ll have to find a yellow stainer in my collection… shouldn’t be too hard!
Try China Glaze Japanese Koi for staining.
Unfortunately I don’t own that one! 🙁
Sinful Colors Unicorn is the worst staining yellow polish I have ever worn. It stains pretty badly even after wearing it for a day or two with a basecoat. Thanks for doing this experiment. After wearing Cult Nails Riot over a peel off basecoat a couple of months ago, I vowed to never wear a peel off base without an actual basecoat on top of it anymore. I’ve just been waiting for the remnants of the staining to grow out, but I tried most of the methods above except cuticle remover at the time.
I’ll have to see if I can get my hands on those for investigating!
Such a cool experiment! I’ve always used cuticle remover for staining followed by a quick swipe (just one or two!) of my buffer if needed. But now I can say “There is evidence this is the best method for staining removal” when I tell my nail friends. I like sounding scientific 🙂
Haha, awesome 🙂
Another excellent post <3
Are dried cuticles a side effect of those harsh treatments?
Not more than usual for me, but I avoided soaking the cuticles by using the cotton pads.
LIFE CHANGING.
I would have never thought to use cuticle remover for staining! Now I can get rid of the leftover staining from Sally hansen ‘The Sky’s the Limit’, my tips are still blue!
Hahaha! Good luck with it 🙂
This is such a great post Michelle! Thank you for making yourself a science experiment for us. I never would have thought that cuticle remover would be a great solution!
People need to be really cautious about buffing though. Here’s an article I wrote explaining why with electron microscope photos provided by Doug Schoon, author of Nail Structure and Product Chemistry. http://www.nailcarehq.com/ridges-in-nails/
Keep up the great work! We need more geeky girls! <3 ~Ana
Definitely! Buffing for me was the easier solution, but for many people carefully rubbing with cuticle remover would be less damaging. Thank you 🙂
About a year ago someone mentioned that Sally Hansen Triple Strong Clear Advanced Gel Nail Fortifier had removed her nail stains. I tried it and it worked! I blabbed to everyone I could and tracked results. About 50% of us got completely clean nails by simply applying 2 coats of Triple Strong as a base under a single mani or 2 (for residual staining). Those that it didn’t work for at least got a useable base out of the deal. I contacted the company, but they weren’t interested. I have no idea why this works, but it does.
Interesting… I don’t think that product is available in Australia though – we miss out on so much here!
Great post!! I need to get me some of that cuticle remover! Thanks for sacrificing your health for us 🙂
A great stainer – and I mean absolutely INTENSE stainer is Zoya – Charla (If you want me to send you a bottle i’d be happy too!)
Also, I don’t know if this topic is worth don’t a separate post on, or if you can answer it here in this comment, but why are white polishes so hard to get right? They always get so gluggy and are hardly ever smooth!
Thanks Michelle!!
It might be the same chromium(III) oxide, since it’s also a teal-ish polish!
I might include that in another random questions post – I’m not totally confident, but I think it’s because you need to pack more pigment in a white polish – it’s just not dark enough to cover everything up unless you do, and then you have too much powder in the polish which leads to chunkiness and glugginess 🙂
great post! my cure for staining is to simply polish over with a darker tone 😛 but thankfully I don’t have too many that do stain! will keep this in mind when I do find trouble 🙂
Haha yes, that was my backup plan!
My suggestion for a yellow stainer is CG Sunshine Pop – it is the polish that turned my nails so yellow that I actually cared to try whitening my nails (I’m generally tolerant of letting stains grow out)… And then the whitening product killed my nails. Ugh. See here for details:
http://www.princesslypolished.com/2013/06/my-horror-story-originails-bubble-white.html
Oh my gosh, that is shockingly bad! I’m so sorry you had to go through that 🙁
Do you get Pure Ice polishes down there? One coat of Wild Thing (http://www.walmart.com/ip/Pure-Ice-Nail-Polish-616-Creme-Wild-Thing-0.5-fl-oz/10422605 … an acid green creme) made my previously-unstained nails ferociously yellow.
Another question: the yellow that comes with this stain seems to be visible only in UV light; in indoor lighting, it’s gone, even for bright lights. I know some neon polishes only “glow” in UV light. What possible pigment does that??
Thank you a million times for your post!!!
I stumbled onto your post, looking for a way to get my horrible stained nails back to normal.
My nails stained pink from Colors by Llarowe Anna’s Adventure.
I had this polish on my nails for 3 days and this is what they looked like after I removed the color:
https://lh6.googleusercontent.com/-8wveN9r9wz0/U9-QX59Yc9I/AAAAAAAAEVw/4hMBY6ExBbU/w440-h330-p/IMG_20140804_122447.jpg
This is the worst case of staining on my nails that I have ever seen!!
I allso found out that this color is no longer available, due to the staining!
I will absolute be trying to get the stains out with Cuticule Remover.
And I hope it will work on pink stains as well!
If you can get your hands on this polish, it is a winner for your study!!
Hi
Silly question but I am looking for products to stain the nails for a week or so. I can’t use nail varnish so need a stain. I have tried food colouring which works really well but the colour is darker near the cuticles even though I have removed them. It leave a u shape boader which is darker that then rest of the nails. Was wondering if you could suggest why and how to stop it. Thanks in advance
I think it’s because that part of your nail is covered by a thin layer of skin – perhaps you could try a cuticle remover?