Skincare fridges are taking off, and the question is – what’s the point of refrigerating your beauty products? Why does cold make them last longer? What about freezing your skincare products – is it OK? The short answer is yes, most of the time. Here’s the longer explanation… (also in video form here!)
Why do beauty products go off?
If you own skincare and makeup products (I assume you all do), you’ll probably have seen a product go off – it might change colour, smell funny, change texture, or grow mould (ew). It might go unnoticed until you put it on your skin and you discover that it suddenly starts breaking you out.
And then there are some products that never seem to go off, like clay powder for mixing masks, or petroleum jelly.
So what’s actually happening when a product goes off?
(a) Ingredients Reacting
Usually, it’s chemical reactions. Most of the time when a product goes off, it’s because the ingredients are undergoing chemical reactions and changing into new substances.
While it might like your product is just sitting there doing nothing, it’s actually really busy at a microscopic level. At any temperature above absolute zero (0 K), which is -273 °C (or -460 °F if you live in a country that hates things that make sense), everything is vibrating with energy.
Heat is a form of energy as well. When you heat something up, all these vibrations intensify.
For a chemical reaction to happen, chemical bonds need to break. This way the atoms are freed and can rearrange and turn into new substances. If there’s a chemical reaction that involves more than one molecule, the two molecules need to hit each other hard enough to break the bonds – otherwise the molecules will just bounce apart without reacting.
In your beauty product, you don’t want reactions to happen. If you have a product with ascorbic acid or vitamin C in it, you (presumably) want it to be vitamin C when you put it on your face.
If you heat up the molecules, they’re vibrating more and moving faster, which means they hit each other harder and more reactions happen faster. On the flip side, we can make the molecules move slower and react slower by cooling them down – which is where your fridge comes in.
(There are a few reactions that don’t involve collisions, but it works almost the same way. Decomposition reactions, where a molecule falls apart, speed up at higher temperatures – if you have a Lego statue and you shake it harder, it’ll break faster.)
(b) Microorganism overgrowth
Even when your product grows bacteria and mould, it’s because of chemical reactions! Every living thing is powered by chemical reactions.
With bacteria and mould, it can be a bit more complex as well. Most organisms have a temperature range where they function and multiply most effectively, and by cooling the product and the microorganisms inside it down, you can get it outside its best range. This is especially effective for “fresh” beauty products that don’t contain preservatives.
(c) Texture changes
One way that products can go off without chemical reactions is when the texture changes.
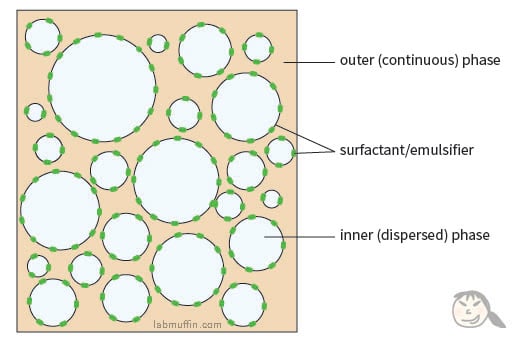
Most beauty products are emulsions which consist of droplets of one thing sitting in another substance (e.g. droplets of oil sitting in water). Oil and water don’t like each other, so there are usually emulsifiers in the product to help the droplets stay in place.
But even with the surfactant, the oil and water would much rather be apart, so they’ll eventually separate. At a higher temperature, there’s more heat, everything moves around more, so the molecules will have more energy to break free of the emulsion structure, and the whole shebang ends up separating faster.
What about freezing?
So if putting your products in the fridge make them go off slower, why not put them in the freezer? Well, a lot of the time, freezing products will actually be better for these reactions. But here’s the catch…
Freezers are cold enough to freeze water, and beauty products contain a lot of water. Freezing has two effects:
Expansion
Water expands when it freezes, especially when you cool it down relatively slowly in a standard freezer. If you have a product that contains whole plant cells, the water will Hulk out and burst the cells, giving you a mushy texture. This happens with things like fresh face masks, and lots of fruit and vegetables.
Coalescence
Water molecules also like to come together or coalesce when they freeze. This means that a lot of the time, when you freeze a product and then defrost it, where the water is located will change.
This is particularly problematic for emulsions. Water freezes from one side of the product to the other. The expanding ice squeezes the oil droplets together, making the emulsion separate, so when you defrost it it won’t have the same texture.
You might have experienced a similar separation if you’ve ever drunken a frozen bottle of drink before it defrosted completely. It starts off super sweet as all the flavour and sugar was squeezed together out of the water, and at the end you’re left with a bland block of ice.
Freezing a product and making the water coalesce can also mess up the preservative system. If the product was formulated properly in the first place, there would be effective levels of preservatives throughout the product. But if the water is moving around, you can end up with pockets of product that are underpreserved and other areas that are overpreserved. The underpreserved areas will grow bacteria far more easily, and since it’s impossible to open a jar without letting bacteria in, that’s bad news for you product’s longevity.
(If you can freeze the product fast enough, expansion and coalescence aren’t an issue because the droplets don’t have time to come together and the ice doesn’t have time to expand as much. That’s why frozen food from the supermarket usually defrosts with a nicer texture than stuff you freeze yourself, and why liquid nitrogen ice cream is so smooth. But this isn’t really possible in your home fridge.)
(There’s also a cool video here from research published in a recent Science paper here that used confocal microscopy to see droplets squeezing together when an emulsion gets frozen slowly.)
Products you really shouldn’t freeze
If you don’t mind the texture change and you just care about keeping the active ingredient molecules intact, and you just defrost enough to use quickly before bacteria and mould can grow, then freezing your products is fine.
For example, Lush Angels on Bare Skin scrub is solid and lasts a few months at room temperature before freezing; after freezing and thawing, it’s much runnier and only lasts a couple of weeks before going mouldy. But since it takes me a year to finish a tub, freezing it in teaspoon sized lumps and thawing a little at a time is the best solution for me!
On the other hand, if there’s a product where texture is really important, then you probably don’t want to freeze it. For example, the microscopic structure of the emulsion is crucial to how well a sunscreen works. Freezing it is going to mess up the structure, and change the concentration of the sunscreen ingredients in different parts of the sunscreen. Foundation is another example where the texture is super important to how well it applies on your skin, so you probably don’t want to freeze it.
What about enzymes?
Some products contain proteins called enzymes, and in some cases enzymes can be denatured and become inactive if frozen. However, the ones most commonly found in skin products – bromelain (pineapple), papain (papaya), actinidain (kiwifruit), fig (ficin) – aren’t significantly affected.
In summary
- Freezing may change the texture of your product, but it’s very unlikely to deactivate the active ingredients.
- This means after it’s thawed, it might separate, be more difficult to apply, be wetter or go mouldy or off faster.
- Freezing small batches separately and thawing them one at a time will make it easier to use. Too much thawing and refreezing should be avoided.
References
CSIRO Collaborative Crystallisation Centre. FAQ – Freezing samples.
Y L Xiong. Protein denaturation and functionality losses. Quality in Frozen Food 1997, 111.
E Jacobsen. Soup or salad? Investigating the action of enzymes in fruit on gelatin. Journal of Chemical Education 1999, 76, 624.






Thanks for writing this post! Now I can freeze all my LUSH face masks without fear 🙂
Yay! Face masks and cleansers are probably the safest to play around with, since they don’t stay for long on your skin 🙂
Thanks for the info!
No worries, hope it comes in handy!
i never thought about freezing skin care products! very interesting!
It’s definitely worth thinking about for natural-based stuff that goes off quickly.
Such a great question and one I would have never thought of! Thanks for such a great explanation! 🙂
Glad you liked it 🙂
Great post. I tend to put things in the fridge and while it doesn’t last as long as freezing at least there’s less chance of separation and I only tend to do this for creamy/watery products.
Fridge is definitely a safer option for stuff where the texture is important. My fridge does this stupid thing though where the cooling isn’t even and half my stuff ends up frozen anyways! Humph.
what type of products can we keep in fridge apart frm sheet masks,eye patches,nd toners????? and can i keep snail cream frm csrx????
You can keep pretty much anything in there! I’d recommend prioritising the ones with unstable actives like vitamin C and vitamin A.
That happens typically when you put stuff too close to the back wall where the actual cooling is (that part freezes) or if the fridge is overfilled. For your fridge to function properly, nothing should touch the back wall, there should be a small gap through where the cold air can circulate. If the air in the fridge can’t move around, uniform temperature can’t establish.
I wish I had a larger fridge so I could refrigerate everything! I usually put stuff in the freezer and forget all about it.
I don’t freeze my skin care stuff, but I put them inside the fridge because I live in a tropical climate.
If I just cool it (no freezing), that would be great right?
Cooling will definitely slow down bacterial growth 🙂
A while ago the lightest shades of Bobbi Brown foundations were taken from the german market(beats me why, there’s so many pale people here) and because it was one of the few pale and nicely formulated products and also easy to get, one girl in my local community had a little freakout. It was her HG foundation and she was like “I needz back ups for da rest of my life!!!1”
I was like ” Yeah, get some, but keep them cool and dark. And keep in mind that they will expire someday so don’t buy more than you will use up within a year or two.”
But now I’m wondering if it was possible to freeze them?
Do you now how silicone based products react to freezing?
I mean, if the texture would drastically change, it would render the foundation practically useless. But a solid cream foundation should be freezeable as opposed to a liquid with high water content.
Also whats with powders etc.?
Maybe a way to preserve vintage make up/ collectors items?
Anyways, thanks for the interesting article, you rock girl!
Greetings, Clarisse
Just to let you know, you can just keep your Angels on Bare Skin face mask in the fridge and it will keep for over a year. I’ve done it twice now and it’s fine. That way I can just scoop out a splodge into a smaller lip balm container that I keep in the shower and use it over a couple of weeks. It’s fine, never gone off.
Hi
I love the explanation.
My questions – how about freezing or refrigerating cosmetic raw materials such as water soluble extract with preservative or emulsifier in pellet form? Would ftidge be better than freezer?
Thanks
Fridge is probably safer, but depending on the material freezer could be better. It’s probably best to ask the supplier!
Hi Michelle, my mom is one that bought backups of backups of her discontinued LIPSTICK (talking about 30+ bullets). Would you recommend keeping them in Fridge/Freezer/Drawer?
I’d recommend somewhere cool – if you have a fridge with space that would be good! A cool storage place should be fine, if you have a cool basement or garage for example.
I never thought about freezing my products, to be honest. I don’t even put them in the fridge, which, following your reasoning, would be advisable (given that the fridge works fine and has an even temperature?
It’s good if your house gets really hot in summer – I personally don’t keep anything in the fridge, but my house stays pretty cool. I also don’t have super unstable products!
Thanks for including Fahrenheit for us backwards types in the US, Myanmar, and Liberia! ?
I keep a LOT of beauty products in the fridge, but especially lipstick and mascara. There’s little as gross as a lipstick whose oils have turned, and I get a lot of mascaras in my Birchbox, so I use one at a time and keep the rest in the fridge.
I also keep powders (eye shadows, blushes) that I seldom use in the fridge. Again, when the oils turn, they smell awful, but by keeping them in the fridge, I can keep ‘em for several years. (No need to wait for these to warm up to use.)
Helpful tip: store nail polish in the fridge. Take it out about an hour before you plan to use it. It lasts waaaay longer this way.
Bonus tip: keep cyanoacrylate (CrazyGlue, SuperGlue, etc) in the freezer. Cyanoacrylate goes bad (thickens, hardens) very quickly after you open the container it’s in, but if you freeze it, that won’t be a problem any longer. Take it out of the freezer before you plan to use it, then return it to the freezer afterwards, and you’ll get waaaay more than just one use out of the tube/bottle.
@Binky Melnik – thanks so much for that tip on SuperGlue! It seems I have to buy a new tube almost every time I’ve opened one (esp since I am prone to breaking things – it’s my HG of household items)!
I totally know what you mean, Ally; I feel the same way. That’s how I know that tip: I’d become frustrated with each tube or bottle hardening nearly immediately, so I asked someone I know who uses it as a finish on wood and polymer. (The latter is yet one more reason I need so much cyanoacrylate: besides using it on broken nails, I’ve begun using it as a finish for polymer as well. It’s as hard as a rock and can be sanded and buffed to a glorious high shine.)
I became frustrated paying good money for higher-end cyanoacrylate, and was horrified that it went bad just as fast as the stuff from the dollar store. Once I started freezing it, no more hardened glue! Yay! So I returned to using the quality stuff again.
(Im like you: if I can touch it, I’ll break it, so finding out how to preserve it made my life a LOT easier!)
I need to buy a mini-fridge for my bathroom! But that means remodeling and I’m not up for that! I have to do my makeup, skin care near a sink – I’m an avid handwasher since I use the back of my hand as if it were an artist palette!